Teena Agrawal* and Priyanka Danai
Banasthali University, Rajasthan, India
Corresponding Author:
Teena Agrawal
Banasthali University, Rajasthan, India
Tel: +91-9680724243
E-mail: tagrawal02@gmail.com
Received date: June 24, 2017; Accepted date: July 03, 2017; Published date: July 07, 2017
Citation: Agrawal T, Danai P. Fossil Early Psilophytian & Lycopodian, Spenopsida Lines of Evolution. Br J Res 2017, 4(3):15. doi: 10.21767/2394-3718.100015
Copyright: © 2017 Agrawal T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Fossils pteridophytes; Evolution; Adaptations; Land plants
Introduction
The pteridophytes formed the dominant part of the vegetation in the historic past. It was the middle of the Paleozoic era when these plants group was well flourished in every place. This flora of the pteridophytes was very abundant till early Mesozoic but late Mesozoic era was well dominated by the gymnosperms. In modern day the pteridophytes flora is replaced by the spermatophytes. These spermatophytes involved the gymnosperms and the angiosperms. Present era is the best flourished by the angiosperms [1,2].
Ages of angiosperms
In whole world the pteridophytes are now of the relict in distribution. They are restricted to the some of the tropical rain forest and the northern hemisphere of the world. However in India the pteridophytes are distributed in to the Himalayas and the Nilgiris. Here large very beautiful tree ferns can be seen with good physiognomy, similarly epiphytic ferns and the other hanging club mosses can be seen in the Nilgiris hills. In India 500 species of the ferns can be seen with different kinds of the pattern of the foliage, which are the taxonomic significance in nature [3-6].
Pteridophytes plants have the long fossil’s history. They have been recognized in the late Silurian period of the Paleozoic era. These plants have the dominant vegetation in whole of the Paleozoic era. The middle and the late Paleozoic era can be regarded as the age of ferns or ages of pteridophytes. The giant lycopsida and the horse tails and the arborescent tree ferns dominated the whole biota at that time. The pteridophytes which are presented by the lycopsida and sphenopsida and the pteridopsida which are of the length of the maximum 6 to 7 feet’s were very abundant in distribution and in height of the trees [7]. They were reportedly around 120 feet’s at the time of the Paleozoic era. They were very abundant and highly s in whole vegetation. This era was the evolution of the pteridophytes and the evolution of the gymnosperms. However in today world they are represented only by the some relict genera and the relict fossils evidences [8].
That era was dominated by the Lepidodendron, sigilalria and the calamites and other fossil lycopsida of that era.
However distribution of the ferns was the matter of the slightly ambiguity, since ferns were of less diversity in the Paleozoic era but as the evolution proceeds and the time passes the diversity of the ferns increase, well a number of the ferns can be seen in that era with great diversity [9].
In India, the fossil pteridophytes are studied by Surange in detail. He has given the whole account of the places in India, which have the long distribution of the fossils pteridophytes. Among the four classes of the pteridophytes like Psilopsida, lycopsida and sphenopsida and pteridopsida, he has described one of the members of psilophyta, seven fossil members of the lycopodiophyta, 12 members of the sphenophyta and 66 members of the pteriphyta. A large number of the fossil ferns have been described in the Rajmahal hills of the India [10]. Bose, Sah and Sharma have described a number of the fossil ferns from the Bihar and other hills of the India. Suthar & Sharma reconstruct the whole solenopteris from the jurrasic flora of the Rajmahal hills.
They found the plant in the form of the leaves, stems, flower, and the seeds in different forms. These plants organs have been termed with different terminology [11-15].
In India the Gondwana land was the point of distribution of the many fossil ferns & horse tails. Glossopteris flora of the Mesozoic was constituted by the many different genera of the sphenophytaof the pteridophyta.
Some the detailed account of the fossils pteridophyta is described below:
Fossil psilophtales: This was the new class which has the assemblages of the fossil plants. This class was coined in the era of the 1917. In that class few genera was included like the Rhynia, horneophyton and psilophytlaes. These plants were distributed during the late Silurian and the Devonian and the Upper Carboniferous period. These plants lack true roots, leaves and the other structures [16].
However developed vascular system can be seen in these groups. Psilophytles are totally different from the other similarly named class termed as the psilotales (Figure 1) [17-20].

Figure 1: Fossil ferns.
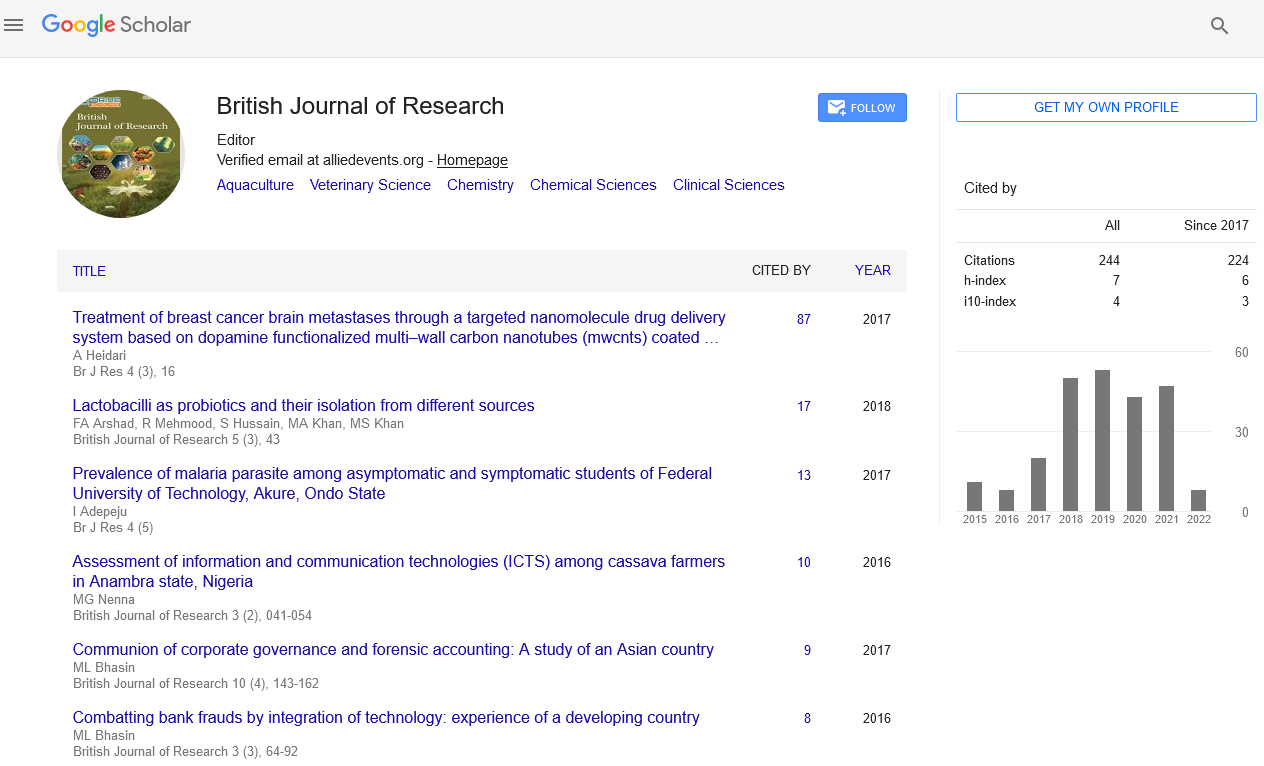
The plant was described by the Kingston and Lang in the rhynia chart of the British islands. Rhynia chert is the place which was formed due to volcanic eruptions. This is the place where a long belt of fossil plants have been observed. The plant was found in different stem, leaves and the isolated sporangia [21]. Typical dichotomous division can be seen in the stems of the Rhynia plants. Horneophyton, is the fossil pteridophytes and it is the linkage between the fossil psilotales and the other living members’ of the spehopsida (Figure 2).
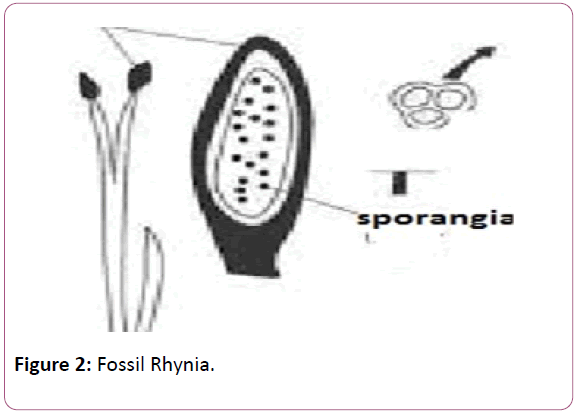
Figure 2: Fossil Rhynia.
These members were also reported form the Rhynia chart of the Scotland in 1920. The sporophytes body is dichotomous in division and the sporangium having the central columella and the other organs of the sores in sides [22].
The presence of the columella in the Hornea is the primitive features of the bryophytes whereas the presence of the tracheids in the sporangia is the presence and the development of the vascular tissue shows the resemblance with the higher plants (Figure 3).
Figure 3: Horneophyton.
Rhynia and horniophyton has been reported form the early Devonian period of the Rhynia Chert of the Scotland. These were the giant and the wet ecosystem of that era.
Fossil lycopsida: The class lycopodiales has the assemblage of the living and the fossil genera’s. This is one of the oldest lineages of the fossil genera’s and the branches [23]. In these fossil genera’s typical heterospory and the alternation of the generation can be seen with clear examples. Some of the fossil genera’s are enlisted in these orders as:
• Lepidodendrales
• Isoetales
• Zosterophyllaeles
Lepidodendrales has the long fossil history, with primitive vascular and arbores cent plants related to the lycopsida [24-26]. This plant group were well flourished in the late carboniferous period, however rapid declines of the lepidodendrales can be seen during the Mesozoic era. These plants group reached to the height up to the 30-40 meters during the devonian and the late Silurian periods (Figure 4).

Figure 4: Reconstruction of the lepidodendrales world.
Lepidodendrales has the long trunk, which reached the height up to the some 40 meters, it was unbranched as well as it bifurcates at the top of the branches [17,20,27]. At the top of the branches one can see the crown of the braches which differentiate it from the rest of the group. Some of the genera’s of the lepidodendrlaes are enlisted as:
• Lepidodendron
• Lepiodpholeois
• Sigillaria
Lepidodendron reproductive structures has-been found in the form of the cones or the separated sporangia [28]. These have been termed as the Lepidostrobus. These cones has always been found in the form of the cones and isolated, not in the form of the attached part of the lepidodendron stem (Figure 5).

Figure 5: Lepidodendrales. A: Fossil reconstruction; B: Fossil image of lepidodendron stem.
Fossils spenopsida: Spenopsida is the class of the assemblage of the fossil and the living pteidophytes, going back to the late Devonian period, these plants commonly known as the horse tails. Living horse tail represent the genus Equisetum [29,30], which are represented by the 20species, these species grows in to the humid areas and generally in to the groups. The vertical stem is represented by the crowns of the leaves, which are present in the alternate sequences of the stem having the ridges’ and the furrows.
Calamitales are the fossils pteridophytes which were arborescent in the Devonian period, whole and the giant forest of the calamitales can be seen in that era. They were the dominant vegetation of the understory of the coal swamps of the carboniferous period [31-36].
A number of the organ taxa have been identified; whole plants are not reported so frequently. Calamites is the name of the fossil stem, other fossil organ genres are enlisted as:
• Arthropitys
• Astromylon
• Annularia
Annularia is the name of the leaves of the calamites stems. They were arranged in the form of the whorls in the group of the 8-11. They were also reported in the form of the separated organs never found attached in anybody [37-39]. Several fragments of the rocks containing prints of fern like foliage. Pteriopsida were found in the non-technical private collection of rocks, and minerals of costarica. The rocks were collected in the rio general valley by personal of aluminium of America. The matrix proved to be the dark grey luciteberlonging to the terraba formations. After the review of the literatures the ferns like organism were found to be the pteropsida (Figure 6).

Figure 6: Fossils leaves of Annularia spehnopsida.
Conclusion
This review article is the assemblages of the some of the discovered early pteridophytes. Most of the fossil pteridophytes have been reported in the Rhynia Chert of Scotland. These are the linkage of the evolution of the early land plants. Since before Pteridophytes the evolution on the land was a basic and significant step, since these pants have a number of the features of the intermediate evolution. Among them fossil psilophytlaes and the lycopodiales, spenopsides presents the lines of evolution of the further spermatophytes of the gymnosperms and angiosperms.
References
- Angiosperm Phylogeny Group [APG III] (2009) An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG III. Botanical Journal of the Linnean Society 161: 105-121.
- Bailey FM (1881) The Fern World of Australia: with homes of the Queensland species. Gordon & Gotch, Brisbane, 105.
- Baker JG (1881) On a collection of ferns made by Kalbreyer W in New Grenada. Trimen's Journal of Botany 10: 202-208.
- Baker JG (1891) A summary of the new ferns which have been discovered or described since 1874. Annals of Botany 5: 455-500.
- Ballard F (1945) A new fern genus from Mexico and Guatemala. American Fern Journal 35: 1-3.
- Beck JB, Windham MD, Yatskievych G, Pryer KM (2010) A diploids-first approach to species delimitation and interpreting polyploid evolution in the fern genus Astrolepis (Pteridaceae). Systematic Botany 35: 223-234.
- Bernhardi DII (1801) Tentamen alterum filices in genera redigendi. Journal für die Botanik (Schrader) 1800(2): 121-136.
- Bierhorst DW (1977) The systematic position of Psilotum and Tmesipteris. Brittonia 29: 3-13.
- Boodle LA (1915) Bulletin of Miscellaneous Information Royal Botanic Garden Kew 1915: 295-296.
- Bower FO (1923) Studies in phylogeny of the Filicales VIII. On Loxsoma and Loxsomopsis. Annals of Botany 37: 349-354.
- Brown R (1810) Prodromus Florae Novae Hollandiae et Insulae Van-Diemen, etc. Leornard Schrag, Nuremberg.
- Brown EDW, Brown FBH (1931) Flora of southeastern Polynesia, II: Pteridophytes. Bernice Pauahi Bishop Museum Bulletin 89: 1-123.
- Brownsey PJ, Lovis JD (1987) Chromosome numbers for the New Zealand species of Psilotum and Tmesipteris, and the phylogenetic relationships of the Psilotales. New Zealand Journal of Botany 25: 439-454.
- Chambers TC (2008) Doodia hindii (Blechnaceae) a new species from north eastern New South Wales, Australia. Telopea 12: 257-261.
- Chase MW, Reveal JL (2009) A phylogenetic classification of the land plants to accompany APG III. Botanical Journal of the Linnean Society 161: 122-127.
- Ching RC (1940) On natural classification of the family "Polypodiaceae". Sunyatsenia 5: 201-268.
- Christ H (1897) Die Farnkräuter der Erde: beschreibende Darstellung der Geschlechter und wichtigeren Arten der Farnpflanzen mit besonderer Berücksichtigung der Exotischen. G. Fischer, Jena.
- Christ H (1904) Primitiae Florae Costaricensis. Filices & Lycopodiaceae III. Bulletin de l'Herbier Boissier ser 1089-1104.
- Christenhusz MJM (2007) Evolutionary history and taxonomy of neotropical marattioid ferns: studies of an ancient lineage of plants. Annales Universitatis Turkuensis ser. AII, vol. 216: 1-134.
- Christenhusz MJM (2009a) Index Pteridophytorum Guadalupensium, a revised checklist of the club mosses and ferns of Guadeloupe (French West Indies). Botanical Journal of the Linnean Society 161: 213-277.
- Christenhusz MJM (2009b) New combinations and an overview of Cyathea subg. Hymenophyllopsis (Cyatheaceae). Phytotaxa 1: 37-42.
- Christenhusz MJM (2010a) Danaea (Marattiaceae) revisited: biodiversity, a new classification and ten new species of a neotropical fern genus. Botanical Journal of the Linnean Society 163: 360-385.
- Christenhusz MJM (2010b) New combinations in the fern genus Tectaria (Tectariaceae) for the Flora of China. Phytotaxa 10: 58-59.
- Christenhusz MJM, Tuomisto H, Metzgar JS, Pryer KM (2008) Evolutionary relationships within the Neotropical, eusporangiate fern genus Danaea (Marattiaceae). Molecular Phylogenetics and Evolution 46: 34-48.
- Christensen C (1906) Index filicum, sive Enumeratio omnium generum specierumque filicum et Hydropteridum ab anno 1753 ad finem anni 1905 descriptorium: adjectis synonymis principalibus, area geographica, etc. H. Hagerup, Copenhagen.
- Christensen C (1910) Ueber einige Farne in O Swartz’ Herbarium. Arkiv för Botanik 9: 1-46.
- Christensen C Skottsberg C (1920) The ferns of Easter Island. The natural history of Juan Fernandez and Easter Island. Almqvist & Wiksells Boktryckeri, Uppsala.
- Churchill H, Tryon R, Barrington DS (1998) Development of the sorus in tree ferns: Dicksoniaceae. Canadian Journal of Botany 76: 1245-1252.
- Colenso W (1881) On some new and undescribed New Zealand Ferns. Transactions of the New Zealand Institute 13: 376-384.
- Conant DS, Raubeson LA, Attwood DK, Stein DB (1995) The relationships of Papuasian Cyatheaceae to New World tree ferns. American Fern Journal 85: 328-340.
- Conant DS, Raubeson LA, Attwood DK, Perera S, Zimmer EA, et al. (1996) Phylogenetic and evolutionary implications of combined analysis of DNA and morphology in the Cyatheaceae. Pteridology in Perspective. Royal Botanic Gardens, Kew.
- Conant DS, Stein DB (2001) Phylogenetic and geographic relationships of the tree ferns (Cyatheaceae) on Mount Kinabalu. Sabah Parks Nat. Journal 4: 25-43.
- Copeland EB (1929) Pteridophyta Novae Caledoniae. University of California Publications in Botany 14: 353-369.
- Copeland EB (1947) Genera Filicum. Chronica Botanica, Waltham, Massachusetts.
- Crabbe JA, Jermy AC, Mickel JT (1975) A new generic sequence for the pteridophyte herbarium. Fern Gazette 11: 141-162.
- Cranfill RB (2001) Phylogenetic Studies in the Polypodiales (Pteridophyta) with an emphasis on the family Blechnaceae. Ph.D. thesis, University of California, Berkeley.
- Cranfill RB, Kato M (2003) Phylogenetics, biogeography and classification of the woodwardioid ferns (Blechnaceae). Pteridology in the New Millennium. Kluwer Academic Publishers, Dordrecht.
- Croft JR (1986) The stipe and rachis vasculature of the dicksonioid fern Cystodium sorbifolium (Cystodiaceae). Kew Bulletin 41: 789-803.
- Degener O (1934) Flora Hawaiiensis or the illustrated flora of the Hawaiian Islands. Honolulu, Hawaii.