Research Article - (2020) Volume 6, Issue 3
Qian Li1, Jie Liu1 and Hui Zhao2
1Department of Integrative Medicine and Neurobiology, Shanghai Medical College, Fudan University, China
2Department of Anesthesiology, Rutgers University, United States.
*Corresponding Author:
Hui Zhao
Department of Anesthesiology
Rutgers University
United States.
Tel: 1-9734247138
E-mail: hz344@scarletmail.rutgers.edu
Received date: April 23, 2020; Accepted date: June 15, 2020; Published date: June 22, 2020
Citation: Li Q, Liu J, Zhao H. FKBP5, A Negative Regulator for Stress Conditioned Alcohol Intake. J Drug Abuse. 2020, 6:3.3
Stressful situations could activate hypothalamic-pituitary-adrenal (HPA) axis, which is relevant to differing tendency to alcohol seeking. In the present experiment, surgical and predator stimulation stresses were undergone, then the mice were tested for alcohol consumption using two-bottle free choice of 10% EtOH and water for 25 days. It was demonstrated that stresses dramatically initiated ethanol intake. During stresses and conditioned ethanol intake, expression of FKBP5 was down-regulated in blood. In the same time, FKBP5 and phosphorylation of glucocorticoid receptor (GR) were down- and up-regulated in prefrontal cortex (PFC) and Para ventricular nucleus (PVN), when over-expression of FKBP5 in both regions could control stresses and the conditioned ethanol intake. Importantly, only in PFC, stresses and ethanol synergistically invoked PKC epsilon translocated into nuclear and enhanced FKBP5 expression machinery including miR-17, 20a, 144, 294, 295, 302, 451. Collectively, we proposed that FKBP5 could pre-sensitize PKC epsilon-dependent gene network in PFC, and prime a hierarchy of signal flow from PFC to PVN. Then, FKBP5 may represent one of biomarkers for post-stress phenotypes including alcohol seeking behavior.
FKBP5, GR, PKC epsilon, Stress, Alcohol intake
Alcohol use disorder (AUD) is a progressive and chronically relapsing disorder, which is characterized by compulsive alcoholseeking, episodes of intoxication, and negative emotional state in the absence of alcohol [1]. Brain systems have been conceptualized as being involved in the progressive deterioration of alcohol control, for example, alcohol-dependent individuals show impairments on PFC-mediated cognitive measures, such as impulse control and reversal learning [2,3]. Also, the rodents chronically exposed to ethanol exhibit deficits in PFC- and PVNmediated tasks, including spatial and reversal learning, setshifting, and fear extinction [4-11]. Then, cortical neuronal circuits and multifactorial adaptation processes might be reorganized by alcohol sensitization. Our previous observation indicated that PFC could integrate the signals of stressful events and the conditioned ethanol consumption [12-15]. Other studies also reported that the sensitized mice drank significant more ethanol than non-sensitized mice; anti-depressant mediated relief of stress-induced behavior was paralleled with counteraction of ethanol consumption [16,17], then stress exposure might trigger ethanol self-administration [18 - 23], the abnormal excitability of neuroanatomical substrate for stress would be part of a complex that is correlated with alcohol related disorders [24-29].
Notably, HPA axis is in fact critical for stress responses [30], reduced GR sensitivity might be the basis for establishment of the interplay with stress and contribute to the negative reinforcement of vulnerability and resilience to alcohol. Based on studies, FK506 binding protein 5 (FKBP5), also referred to as FKBP51, was originally identified as component of the progesterone receptor-chaperone heterocomplex [31-35], which could influence GR sensitivity to cortisol and efficiency of negative feedback on the HPA axis. Moreover, FKBP5 was one of the most important associations with stress-related phenotypes including depression and posttraumatic stress disorder [36-39], some of FKBP5 risk alleles were found to be linked to alcohol consumption phenotypes in mice and humans [40-42]. Therefore, we hypothesized that FKBP5 might reprogram PFC neuronal circuits, compromise their interplay with HPA axis and integrate signals in response to stressful events and the conditioned behavioral phenotypes.
Stress animal model
Male mice (Animal Center of Chinese Academy of Sciences, 18–20 g) were individually housed in controlled environment with 12h light–dark cycle and allowed to acclimate for minimum of 7 days. All experimental procedures were carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
For traumatic stress, after anesthetized with isoflurane, mice were incised longitudinally to a length of 3 cm along the dorsal median line and 3 cm along the abdominal median line. Five minutes after surgery, the wounds were sutured, animals were kept warm under standard housing conditions, no post-operative infection occurred [43]. For PSS, mice were in contact with a cloth with the smell of cat urine for 14 days, efforts were made to minimize the numbers of animal used and their suffering.
Microinjection
After anesthesia, mice were bilaterally implanted with guide cannulae (outer diameter, 0.41 mm; inner diameter, 0.25 mm; RWD Life Science, Shenzhen, China). Cannulae was inserted into PFC (AP: +1.75 mm; L: ±0.75 mm; V: −2.65 mm) and PVN (AP: −0.82 mm; L: ± 0.5 mm; V: −2.0 mm) based on Stereotaxic coordinates of mouse brain. Microinjection was performed at a rate of 0.25 μl/min for 4 min, cannula was kept for an additional 2 min at the end of injection, and injection site was assessed under light microscopy.
Ethanol consumption
Mice were individually housed, each cage was outfitted with two drinking bottles made of graduated serological pipettes attached to ball bearing sipper tubes and plugged with silicone stoppers. Both bottles were filled with water for three days, then one each with water or 10% ethanol for up to 25 days, which were secured to the wires of a normal cage insert using binder clips and joints were sealed with Para film to reduce evaporation. The position of the bottles was alternated every day relative to the cage walls to avoid any place preference bias. Volume levels were recorded at noon to determine daily consumption and subsequently tubes were refilled with the appropriate solution.
Mice were weighed each day and the value was used to calculate individual ethanol concentrations over a 24h period. Ethanol intake was calculated by dividing the amount of ethanolcontaining solution consumed by total fluid intake. Mice were also tested for two-bottle choice of saccharin (0.03%, 0.06%) or quinine (0.015, 0.03 and 0.06 mM) consumption for 25 days.
Recombinant adenovirus construction
Recombinant adenovirus expressing mice FKBP5 was constructed by inserting into adenoviral shuttle vector pDE1sp1A (Microbix Biosystems, Inc. Canada), the insert was then switched to the adenoviral vector through LR recombination. Adenovirus was puriÃÆïÃâìÃâÃÂed by CsCl2 gradients and PD-10 Sephadex chromatography. After homologous recombination with the backbone vector PJM17, plaques resulting from viral cytopathic effects were selected and expanded in 293 cells.
Blood ethanol concentration (BEC)
After 25 days of alcohol intake, blood samples were taken and collected into a sterile microcentrifuge tube from facial vein by making submandibular puncture with 18 1/2 gauge needle tip. Two 20 μl volumes were then pipetted immediately into either ZnSO4 or Ringer solution for subsequent assays. When assessed by gas chromatography (GC), 20 μl of blood was transferred into a tube with 50 μl of cold ZnSO4 solution, subsequently with addition of 50 μl of 0.3 N Ba(OH)2 and 300 μl deionized water to each tube. Samples were then vortex for 10 s and spun down for 5 min at 12,000 RPM in a 4 °C centrifuge, 350 μl of supernatant was drawn off into a GC vial and processed through an Agilent 6890N gas chromatograph with attached 7683 autosampler and a J&W scientific 30 m × 0.53 mm column. Detector output was quantified by using Chemstation software data were calculated relative to standards.
RNA isolation and real-time PCR
Total RNA was extracted from brain slices using Direct-sol MiniPrep kit (Zymo Research) according to the manufacturer's instructions. For each sample, RNA quantification and quality control were assessed by NanoDrop 2000c measurements (Thermo Scientific), a final amount of 5 ng of the template RNA was required for first-strand cDNA synthesis of miRNAs with the Universal cDNA Synthesis kit II (Exiqon). Real-Time PCRs was carried out on ABI 7500 Fast Sequence Detection System (Applied Biosystems), with protocol of 40 cycles with denaturation at 95°C for 15 seconds, annealing and extension at 60°C for 1 minute. Primers were listed in Table 1, beta-actin was used to normalize expression level of miRs.
Table 1 Primer list 5’-3’
Immunofluorescent labeling
Mice were anesthetized with isoflurane and transcardially exsanguinated with 0.1M PBS followed by perfusion of the fixation (4% paraformaldehyde in 0.1M PBS, pH7.4), each provided in a 7 ml/min flow rate. Serial sets of 20 μm coronal brain sections were collected on a freezing microstome (Leica, SM2000R). Frozen sections were subjected to anti-FKBP5 (1:1000, ab46002) Abcam, Cambridge, CB) or anti-GR (phospho S211/S226) antibodies (1;1000, Abcam, ab55189/ab93104), and Alexa Fluor 594- or 488-conjugated secondary antibody (1:500, Invitrogen, Carlsbad, CA). Data derived from each group were analyzed by Leika Q500IW image analysis system. For statistical analysis, fluorescent density is reported as the average density of all individual animals ± SEM in each experimental group (n = 5). The mean density of all the microphotographs was analyzed with the aid of ImageJ analysis software.
Subcellular fractionation
Nuclear extracts were prepared from PFC. Briefly, 1ml of extraction buffer was added (10 mM Hepes, pH7.9, 1.5 mM MgCl2, 10 mM KCl) together with the recommended amount of protease inhibitor cocktails. After three freeze-thaw cycles, cytoplasmic extracts were recovered by centrifugation at 15,000 × g for 5 min, and the pellets were resuspended in buffer C (20 mM Hepes, pH7.9, 1.5 mM MgCl2, 420 mM KCl, 0.2mM EDTA, 25% glycerol) together with the recommended amount of protease inhibitor cocktails. Following a 30min incubation at 4°C, nuclear extracts were recovered by centrifugation at 15,000 × g for 5 min.
Western Blotting and ELISA
Brain tissue was dissected and homogenized in ice-cold lysis buffer containing 137 mM NaCl, 10 mM Tris-HCl, pH 8.0, 1 mM EDTA, pH 8.0, 1% NP-40, 10% glycerol, 1 mM phenylmethylsulphonyl fluoride, 10 mg/ml aprotinin, 1mg/ml leupeptin, and 0.5 mM sodium vanadate. The tissue homogenate solutions were centrifuged at 14,000 × g for 5 min at 4°C. The supernatants were collected and used for quantification of total protein. For Western Blotting, proteins were resolved in SDSpolyacrylamide gel, and transferred to polyvinylidene difluoride membrane (GE Healthcare, Piscataway, NY). The membrane was probed in the presence of anti-PKC epsilon (1:1000; BD Transduction Laboratories, Lexington, KY). Then the membrane was incubated with secondary antibody conjugated with alkaline phosphatase, protein bands were detected by ECF substrate and scanned in the Storm 860 Imaging System. The band intensities were quantified and analyzed with the Image Quant software (GE Healthcare). For ELISA, levels of FKBP5 and pGR were assessed using a commercially available assay kit from Bio compare, color change was measured in an ELISA plate reader at 450 nm.
Brain slices cultures
Mice were euthanized by decapitation, and the brains were rapidly removed into ice-cold dissection buffer (2.6 mM KCl, 3 mM MgCl2, 1 mM CaCl2, 1.23 mM NaH2PO4, 10 mM dextrose, 212.7 mM sucrose, and 26 mM NaHCO3). Acute coronal slices (400 μm) were prepared from PFC/PVN and transferred to a holding chamber containing artificial cerebrospinal fluid (124 mM NaCl, 5 mM KCl, 1.25 mM NaH2PO4, 10 mM dextrose, 1.5 mM MgCl2, 2.5 mM CaCl2, and 26 mM NaHCO3) bubbled with 95% O2, 5% CO2 maintained at 32°C. Slices were recovered for minimum of 90 min before overnight incubation, then exposure to the indicated treatment.
Data analysis
This experiment was performed independently with the same parameters and normalized results were pooled. For immunofluorescent analysis, three adjacent slices from each animal were stained and quantified. ANOVA (one/two-way) were performed using GraphPad Prism 5 software to analyze difference among means of normalized results. Differences with P-value less than 0.05 were considered statistically significant.
Effect of stress on operant ethanol intake
Mice were divided into 5 groups: control, trauma 1d, trauma 3d and trauma 7d, and PSS, with 6 animals in each group, then ethanol (10 %) intake was recorded using two-bottle choice paradigm over 30 days (the timeline is described in Figure 1A, In Figure 2A and Figure 2B illustrated that, in groups of trauma 1d and 3d, not in control or trauma 7d, a tendency toward ethanol consumption occurred on 14-20 days, with evident ethanol preference over saccharin and quinine. Comparably, ethanol consumption occurred a little earlier in PSS mice (10-20 days) than the counterparts under surgical stress.
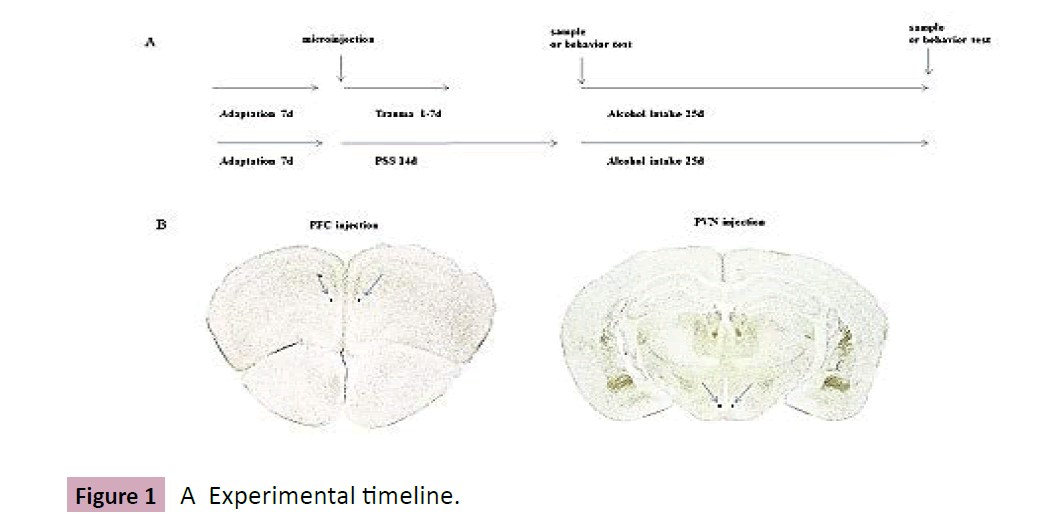
Figure 1: A Experimental timeline.
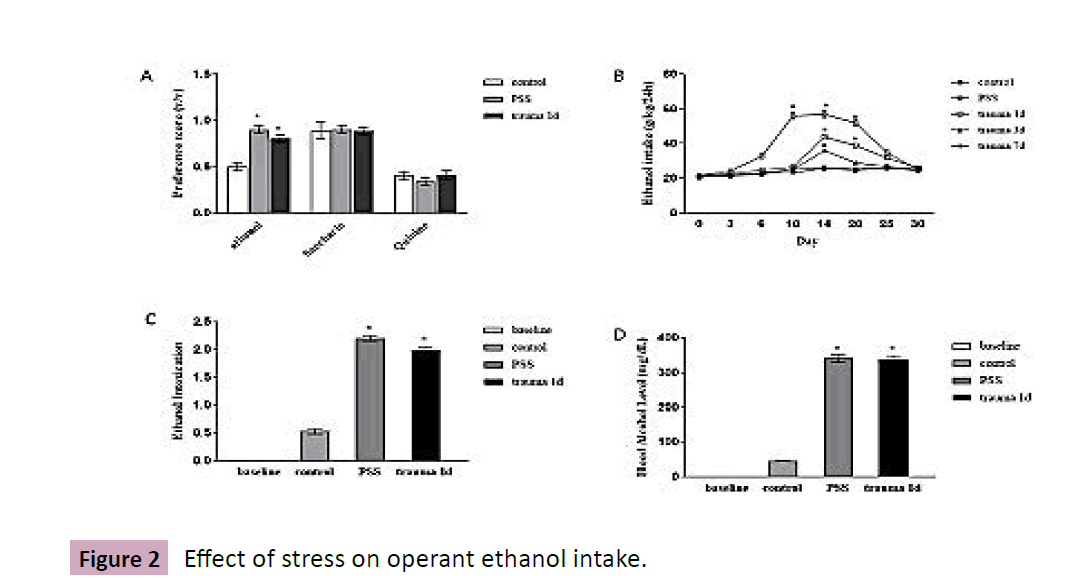
Figure 2:Effect of stress on operant ethanol intake.
A, Experimental timeline. Mice are adapted in animal room for 7 days, then divided into several groups as indicated in Results. For microinjection, 24 or 72 hours after injected with PKC epsilon inhibitor or FKBP5 virus, animals are undergone stresses and tested with ethanol consumption procedure using two-bottle choice. B, Identification of stereotaxic micro-injection site, the coordinates are indicated in Materials and methods (Figure 1A).
To confirm ethanol intake, ethanol intoxication behavioral and blood ethanol concentrations were detected. As shown in Figure 2C, by using six-point intoxication scale (0, normal mice; 1, hypoactive; 2, ataxia; 3, ataxia with dragging abdomen; 4, loss of righting reflexes; 5, loss of eye blink reflex), stressed mice displayed a full range of intoxication after ethanol intake (F=778.7, P<0.001). Also, blood ethanol concentration in PSS and traumatic mice were significantly increased compared with non-stress group, which were in the range of 341.2±23.9 and 338.4±21.7 mg/DL, respectively.
Mice are undergone traumatic or PSS stimulation, respectively, then are tested with ethanol consumption procedure using twobottle choice. A, Ethanol preference is tested for two-bottle choice with saccharin and quinine, consumption is calculated by dividing the amount of ethanol-containing solution by total fluid intake; B, Mice are weighed each day and this value is used to calculate individual ethanol concentrations over a 24-h period; C, a six-point behavioral intoxication scale (0, normal rats; 1, hypoactive; 2, ataxia; 3, ataxia with dragging abdomen; 4, loss of righting reflexes; 5, loss of eye blink reflex) was assessed; D, Blood samples are collected, BEC assay is used to measure blood alcohol levels. Data are normalized and calculated, each value represents mean ± S.E.M *p < 0.05 vs control. (Figure 2).
FKBP5-GR signaling in the stress conditioned ethanol intake
Mice were divided into 3 groups: control, trauma 1d, and PSS, with 12 animals in each group, and subdivided into saline and ethanol groups. Immunofluorescent analysis in PFC was shown in Figure and relative to water-drinking controls, chronic ethanol consumption produced around 60% decrease in FKBP5 expression in PFC and PVN in stressed mice. By ELISA assay, blood concentration of FKBP displayed similar change pattern, was down-regulated during stresses and the conditioned ethanol consumption. . Mice are undergone traumatic or PSS stimulation, respectively, then are tested with ethanol consumption procedure using two-bottle choice. A, Brain slice was prepared, expressions of FKBP5 and pGR in PVN are detected by Immunofluorescent analysis. B, depicts the expression of FKBP5 in hippocampus, PVN; expression in blood is detected by ELISA assay; C, depicts pGR expression in hippocampus and PVN. Data are normalized and calculated as percentage of control, each value represents mean ± S.E.M *p < 0.05 vs control. Scale bar = 50 cm. (Figure.3)
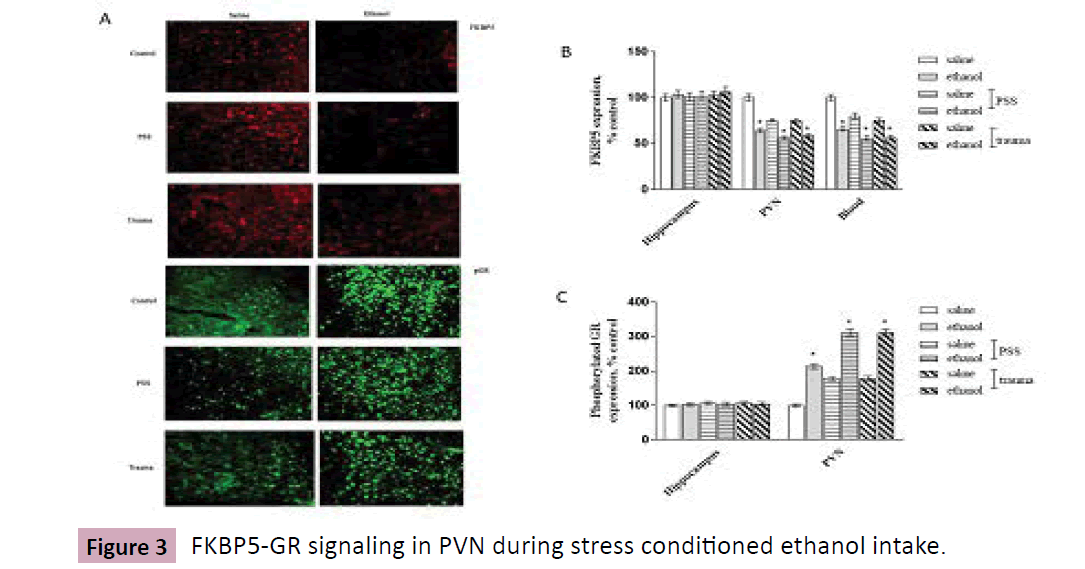
Figure 3:FKBP5-GR signaling in PVN during stress conditioned ethanol intake.
We further examined GR phosphorylation in PFC and PVN during stresses and the conditioned ethanol consumption. As illustrated (Figure 3-5), stresses and ethanol induced an apparent elevation in GR phosphor-protein levels, with a significant stress x ethanol correlation.
Figure 4:HE staining of brain slice. A, PFC; B, PVN.
Mice are undergone traumatic or PSS stimulation, respectively, then are tested with ethanol consumption procedure using twobottle choice. A and C, brain slice was prepared, expressions of FKBP5 and pGR in PFC are detected by Immunohistochemistry; B and D, depict the related data analysis. Data are normalized and calculated as percentage of control, each value represents mean ± S.E.M *p < 0.05 vs control. Scale bar = 50 cm. (Figure 5)
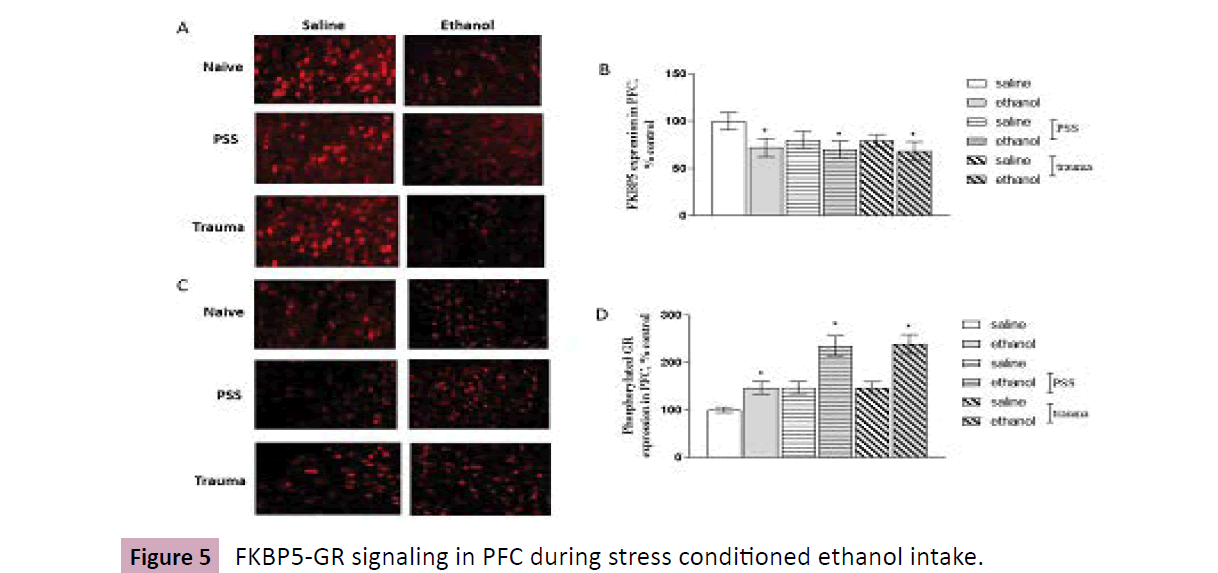
Figure 5:FKBP5-GR signaling in PFC during stress conditioned ethanol intake.
Over-expression of FKBP5 on the stress conditioned ethanol intake
Mice were injection of adenovirus expressing FKBP5 into PFC and PVN, then were undergone stresses, and ethanol (10%) intake. By Western Blot analysis, Figure 6A confirmed that FKBP was over-expressed in PFC and PVN after injection. Ethanol intake was subsequently recorded, as shown in Figure 6 B-E, after Mock injection, stressed mice had more and global increase in ethanol consumption than the control group. Comparably, overexpression of FKBP5 completely blocked ethanol intake both in non-stressed and stressed mice.
Mice are undergone traumatic or PSS stimulation, in the presence or absence of FKBP5 into PFC/PVN, then tested with ethanol consumption procedure using two-bottle choice. A, FKBP5 expression in PFC and PVN were detected by Western Blot analysis. B and D, mice are weighed each day and this value is used to calculate individual ethanol concentrations over a 24-h period. C and E, amount of ethanol consumption is calculated over 25 days of total intake. Data are normalized and calculated as percentage of control, each value represents mean ± S.E.M *p < 0.05 vs control (Figure 6).
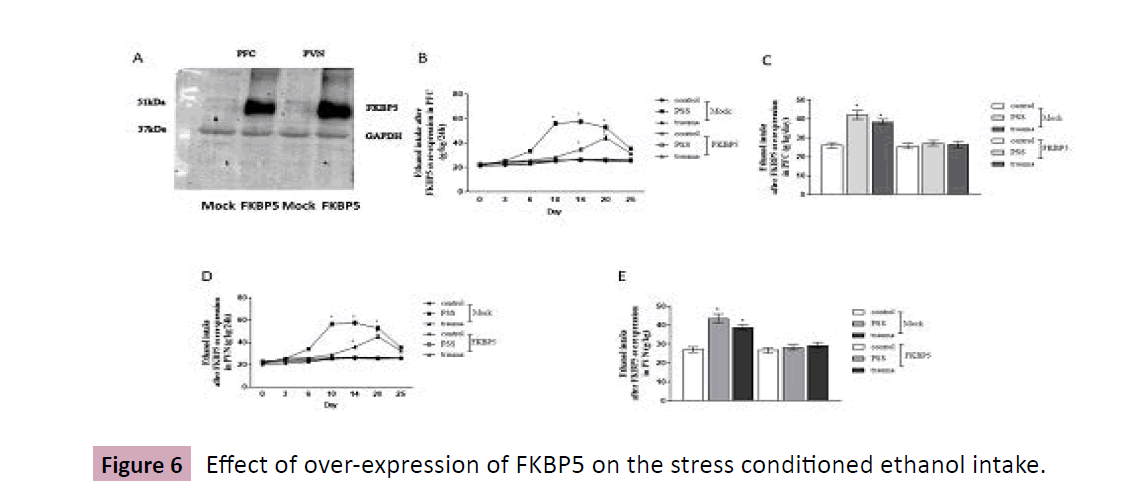
Figure 6:Effect of over-expression of FKBP5 on the stress conditioned ethanol intake.
Protein kinases in the FKBP5-pGR signaling during stresses conditioned ethanol intake
Brain slices from PFC and PVN were cultured, expressions of FKBP5 and Phosphor-GR expression were detected by ELISA assay. It was shown in Figure 7A and Figure 7B that, after incubation with serine/threonine kinase inhibitors PD98059 (ERK), LY294002 (Akt), and GF109203X (PKC) for the indicated time, ethanol evoked down- and up-regulation of FKBP5 (F = 66.43, P<0.0001) and pGR (F = 68.71, P<0.0001) in PFC and PVN were partially inhibited by PD98059 and LY294002, but the alterations in PFC was completely reversed by GF109203X.
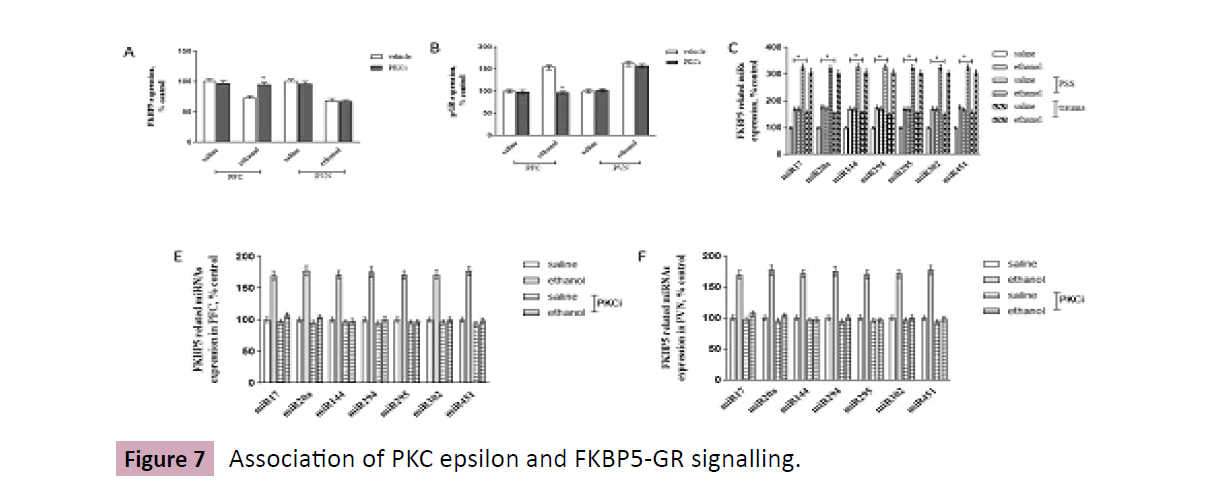
Figure 7:Association of PKC epsilon and FKBP5-GR signalling.
After literature survey, several FKBP5 related microRNAs (43) were detected by real-time PCR, it was revealed that, compared with control mice, stressed mice displayed significant initiation of miR-17, 20a, 144, 294, 295, 302, 451 in PFC, a further 50% induction after exposure to ethanol (Two ANOVA analysis: Factor = 95.84, P < 0.001) (Figure 7C). By using brain slice culture from PFC, real-time PCR data illustrated that, expressions of FKBP5 related micro-RNAs (miR-17, 20a, 144, 294, 295, 302, 451) was initiated by ethanol exposure, the effect could be inhibited in the presence of PKC epsilon inhibitor peptide (10mM) (F = 1569, P < 0.0001, Figure 7D).
A and B, Brain slices from PFC and PVN were cultured, in the presence or absence of protein inhibitors, PD98059, LY291004, GF109203X, expressions of FKBP5 and Phosphor-GR expression were detected by ELISA assay; C, Mice are undergone traumatic or PSS stimulation, then are tested with ethanol consumption procedure using two-bottle choice. Expressions of FKBP5 related micro-RNAs in PFC are analyzed by real-time PCR; D, Brain slices from PFC were cultured, in the presence or absence of PKC inhibition peptide, expressions of FKBP5 related micro-RNAs are analyzed by real-time PCR. Data are normalized and calculated as percentage of control, each value represents mean ± S.E.M *p < 0.05 vs saline (Figure 7).
PKC epsilon in the KBP5-GR signaling stress conditioned ethanol intake
Mice were undergone stresses and ethanol intake, Western Blot analysis demonstrated in Figure 8A and B that, in the presence of stresses, nuclear expression of PKC epsilon in PFC were increased around 1.9 - 2.1 folds relative to baseline, which was further strengthened by 1.4-1.5 folds following ethanol exposure (F = 133.0, P < 0.0001).
Mice were divided into 2 groups, with 18 in each group, all animals were implanted cannulae, PFC/PVN injection of FKBP5 with or without PKC epsilon inhibitor peptide (0.4 mg/kg; Santa Cruz, sc3095). 72h later, animals were subdivided into 3 groups: control, trauma 1d, and PSS, with ethanol (10 %) intake was recorded using two-bottle choice paradigm over 25 days. Figure 8C illustrated that the global increase in ethanol intake in stressed mice was completely blocked by FKBP5, but was interrupted by PKC epsilon inhibition in PFC (F = 5.059, P < 0.001), but not in PVN.
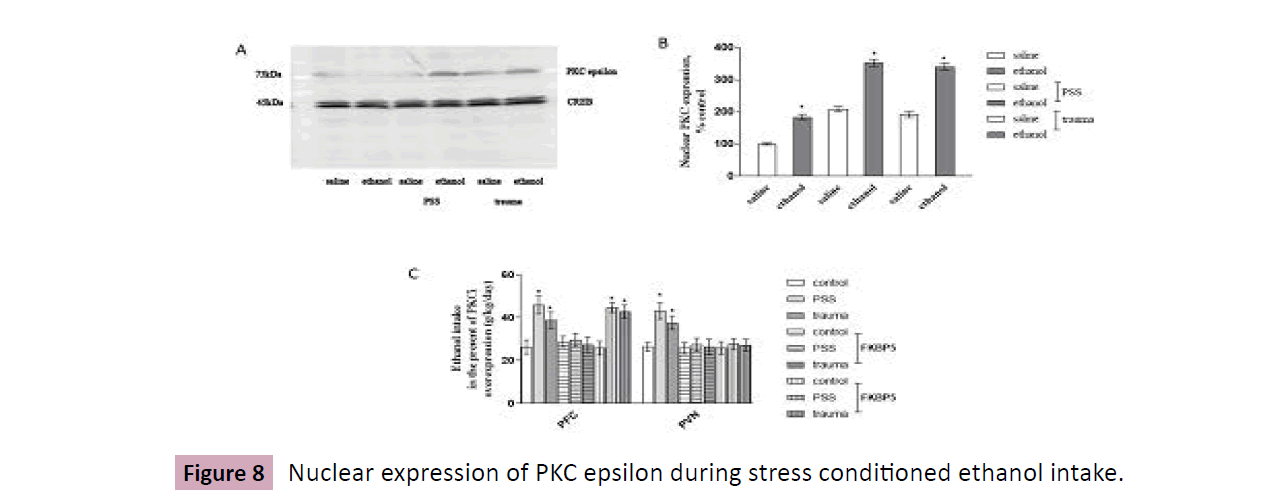
Figure 8:Nuclear expression of PKC epsilon during stress conditioned ethanol intake.
Mice with PFC or PVN injection of adeno-virus FKBP5, are undergone traumatic or PSS stimulation, respectively, then are tested with ethanol consumption procedure using two-bottle choice. A and B, Nuclear fraction is separated from PFC, PKC epsilon is detected by Western Blot analysis, data are normalized and calculated as percentage of control, each value represents mean ± S.E.M of five independent experiments. *p < 0.05 vs saline. C, Mice are undergone traumatic or PSS stimulation, in the presence or absence of PKC epsilon inhibition peptide (PFC injection), then are outfitted with ethanol consumption procedure using two-bottle choice. Mice are weighed each day and this value is used to calculate individual ethanol concentrations over a 24-h period. Data are normalized and calculated as percentage of control, each value represents mean ± S.E.M *p < 0.05 vs control. (Figure 8).
It is reported that the transition from casual drinking to alcohol dependence involves numerous neuroadaptive changes in brain stress systems [44]. Postmortem studies confirmed that structural and molecular alterations in PFC are related with chronic alcohol abuse or dependence. Our present data provided direct evidence that stresses invoked ethanol intake, which strengthened the idea that recruitment of brain stress systems prime the aversive aspects of alcohol seeking behaviors.
Over the decades, multiple studies accumulated for FKBP5, a static factor that calibrates GR activity via the ultra-short intracellular feedback loop, abnormal GR phosphorylation was link to stress and post-traumatic stress disorder [45]. For drinking behavior, our and other studies demonstrated that that FKBP5 in PFC, PVN, nucleus accumbens and ventral tegmental area was diminished during stress conditioned ethanol seeking [41,42]. Moreover, we found that over-expression of FKBP5 in PFC or PVN could prevent stresses and the conditioned ethanol drinking. In parallel with the inhibition in brain level, blood FKBP5 was decreased, accompanied with elevated phosphor-GR in PFC and PVN. Then, FKBP5 might reprogram GR neuronal circuit and establish a distinct signal pathway in PFC and PVN during stressful events.
As shown in our present experiment that enhanced phosphorylation of GR occurred at serine 221 or 226, the potential locations related to JNK [46]. Our previous study indicated that PKC-JNK signal cascades in PFC were correlated with stressful events [47-49], together with the inversely modulation of FKBP5 and GR phosphorylation during stress conditioned ethanol intake, the data suggested that FKBP5 might create a micro-environment in PFC that sense and control stress responses. To prove that anatomical organization of PFC and PVN was a critical factor for stress coping capability, we used brain slice culture from PFC and PVN, data showed that PKC epsilon in PFC was more essential for stressful information flow, which could organize FKBP5 gene networks via micro-RNAs (miR-17, 20a, 144, 294, 295, 302, 451). In addition, PKC epsilon was enriched in nuclear during stress conditioned ethanol intake, inhibition of PKC epsilon in PFC not PVN could interrupt FKBP5’ blockade on the output of stressful event. Therefore, present data indicated that PKC epsilon was recruited into FKBP5 regulatory machine in response to stress, and served as a potential driving factor for signal flow from PFC to PVN. Since FKBP5 production in brain and blood was in parallel inhibited following stress and the conditioned ethanol intake, which could be used for reflecting outcome of stress and the conditioned ethanol intake.
We detected a significant initiation of ethanol intake following traumatic and psychological stresses. PKC epsilon sensitized FKBP5 gene networks in PFC, which might prime the hierarchy for stress signaling flow from PFC to PVN. Therefore, FKBP5 has emerged as one of the most intriguing associations with stressrelated phenotypes, and may represent one of biomarkers for stress conditioned alcohol seeking behavior.
HZ produced the hypothesis for this study and was responsible for the manuscript preparation. QL and JL were responsible for the molecular, behavioral study. All authors read the manuscript, approved the final draft and declare that they have no competing interests.
The study was supported by National Nature Sciences of China (81471370, 81573712) and National Key Basic Research Program of China (2013CB531900).