Keywords
Silver nanoparticles (AgNPs); MagPI; Biofilm adhesion; Biofilm adhesiveness; Mono-species biofilm
Introduction
Formation, growth and development of biofilms are decisively influenced by their adhesiveness because this parameter is essential for the overall stability and structure of biofilms. This is of very high importance, as biofilms provide valuable ecosystem services on the landscape scale, e.g. the stabilization of riverine fine sediments acting also as sinks for pollutants and contaminants [1-4]. On the other hand, in several cases formation of biofilms is regarded adverse as it constitutes significant economic and health hazards e.g. on heat exchangers, hulks or implants [5-7]. This ambivalence of biofilm formation is a reason why different modelling approaches which allow simulation and predictions of expected biofilm development, are currently receiving steadily increasing scientific attention [8-10]. In this context, a variety of impact factors on biofilm development and stability have been studied such as the impact of different biotic or abiotic boundary conditions or the effect of an exposure to herbicides or antibiotics [11-14].
Nanoparticles in general and silver nanoparticles (AgNPs) in particular, constitute a rather new class of emerging substances whose environmental effects are currently subject of intensive research. A reason for this current scientific interest is the fact that AgNPs contribute a major fraction to all produced nanoparticles [15] added to various products of daily life due to their antimicrobial effects [15]. Furthermore, AgNPs could be shown to impact benthic biofilms in various ways [16-18]. However, analyses of the impact of AgNPs on the structure and stability of biofilms such as the one presented by Grün et al. are rare [19]. In fact, the presented experiments with mono-species biofilms of the model bacterium Aquabacterium citratiphilum process high significance for natural benthic biofilm systems. In natural complex biofilms, the symbiosis between diatoms and bacteria was reported to be essential for biofilm stability. Lubarsky et al. could indicate the crucial role of a mixed-taxa symbiosis for biostabilization as biofilm assemblages consisting of bacteria as well as diatoms displayed higher stability than pure bacterial or axenic diatom biofilms [3]. Furthermore, Aquabacterium citratiphilum was described as ubiquitous freshwater microbe with a specialized metabolism with an increased capacity to utilize exudates of diatoms [20,21]. These findings suggest a possible role of this species in a symbiotic relationship with diatoms. The structural relevance of this symbiosis between diatoms and their associated satellite bacteria was further described by Windler et al. [22]. As a reaction of the activity of satellite bacteria, diatoms of the species Achnanthidium minutissimum modified their EPS production to form a mucilage capsule and changed from a planktonic state to a lifestyle attached to the substrate. This reaction may have a direct impact on biostabilization because a permanent attachment of microbes to sediment grains coupled with high EPS production can constitute one important trigger for the stabilization of the colonized fine sediment. These observations and the fact that Aquabacterium citratiphilum was identified as a dominant bacterial species in natural lotic biofilms (Schmidt et al. [12], unpublished) suggest its role as a diatom satellite bacterium with potential high structural importance for the overall system of natural complex benthic biofilms.
The aim of this study was to expand the work with the model bacterium Aquabacterium citratiphilum and perform an up to now unique comprehensive analysis of the impact of an exposure to AgNPs on biofilm structure and stability. A treatment with an environmental relevant, low-dose concentration of AgNPs that was shown to impact the structure and mechanical stability of freshwater bacterial biofilms was compared with untreated controls [19]. Furthermore, three positive controls exposed to higher concentrations of AgNPs, ionic silver (AgNO3) and copper sulphate (CuSO4) with expected clear negative impact on the vitality of the bacteria and the stability of the biofilm system were included. The comprehensive analysis approach of this study incorporates the investigation of the composition of extracellular polymeric substances (EPS), the biofilm structure (via epifluorescence microscopy) and the resulting overall functionality of the biofilm system (adhesiveness). For the assessment of biofilm adhesiveness, an enhanced magnetic particle induction (MagPI) device was utilized [23]. "This study is the first to modify this device that was originally designed for an investigation of the stability of intertidal biofilms to assess the adhesiveness of the described model biofilms [3]." By a series of modification and optimization, the sensitivity and reproducibility of this method could be significantly enhanced for an assessment of even small changes in biofilm adhesiveness. One focus of this study was to utilize this optimized technique to investigate the impact of an exposure to citrate-coated AgNPs on model freshwater biofilms. Thus, this study is the first to address the potential adverse effects of an exposure to AgNPs on biofilm adhesiveness by an extended series of MagPI measurements.
Materials and Methods
Experimental design
Aquabacterium citratiphilum 20 (DSM 11900, DSMZ, German Collection of Microorganisms and Cell Cultures, Germany) was cultivated in the modified R2A medium (DSMZ 830a) with constant agitation for 72 h at 21°C. 100 ml of this preculture representing a mean biomass of 2.6 ± 0.3 × 107 cells/ml were thoroughly mixed with 4 L fresh modified R2A medium. For biofilm growth, aseptic Petri dishes loaded with two Poly-L-lysine coated glass slides each were filled with 100 ml of the Aquabacterium cell suspension and incubated for 120 h at 21°C. Biofilm samples were divided into four subsets as displayed in (C, T1/T2, T3, T4) of 25 glass slides each. Test dispersions of AgNPs and solutions of AgNO3 or CuSO4 were thoroughly mixed with the medium up to the final concentrations of 600 μg l-1 and 2.4 mg l-1 for AgNPs, 2.4 mgl-1 for AgNO3 and 125 mgl-1 for CuSO4. MagPI measurements were performed on three different random spots on each single slide after 24 h exposure and on the control biofilms (Table 1).
| Set |
Phase 1 |
Measurement (after 24 h incubation) |
Phase 2 |
Measurement (after 24 h incubation) |
| C |
- |
MagPI, EPS |
- |
MagPI, EPS |
| T1* |
600 µg/l AgNP |
MagPI, EPS |
- |
- |
| T2* |
- |
- |
2.4 mg/l AgNP |
MagPI, EPS |
| T3 |
- |
- |
2.4 mg/l AgNO3 |
MagPI, EPS |
| T4 |
- |
- |
125 mg/l CuSO4 |
MagPI, EPS |
*Treatments were performed subsequently on the same glass slides in the same petri dishes.
Table 1 Experimental proceeding.
Silver nanoparticles (AgNPs)
The citrate-coated AgNPs applied in this study were synthesized and characterized as previously described by Metreveli et al. [24]. A modified citrate reduction method [25] was used to synthesize the applied AgNPs with a diameter of 30 nm [25]. For this purpose, 5 mmol of Na3C6H5O7 (Sigma-Aldrich, Germany) and 1 mmol AgNO3 (AgNO3, ≥ 99.9%, p.a., Carl Roth, Germany) were dissolved in 950 ml and 50 ml deionized (Milli-Q) water respectively. After adjustment of pH 11 using 1 mol l-1 NaOH (Carl Roth, Germany), the trisodium citrate solution was boiled under permanent stirring (RCT basic, IKA Labortechnik). Within 5 min, the silver nitrate solution was added drop wise to the boiling trisodium citrate solution and continuously stirred and boiled for 20 min until no changes in the color could be detected. Subsequently, the nanoparticle dispersion was cooled to room temperature, and filled up to 1 L with deionized water to compensate evaporation losses. The final pH value in the dispersion was 8.9. The synthesized AgNP stock dispersion was stored in the dark at 4°C.
The hydrodynamic diameter of the AgNPs was determined by dynamic light scattering (DLS) with a scattering angle of 165°. The zeta potential was measured utilizing the electrophoretic light scattering technique followed be subsequent calculation using the Smoluchowski equation (both using Delsa Nano C, Beckman Coulter). For the DLS and zeta potential measurements the stock dispersion of AgNPs was diluted with demineralized (Milli-Q) water by a factor of 1:3.
Adhesiveness of the biofilm surface
The adhesiveness of the biofilms was determined on three different, randomly selected spots on 25 biofilm covered glass slides per treatment and control, resulting in 25 biological replicates. For this purpose, a MagPI (magnetic particle induction) system was used that was enhanced in a series of modifications [23]. The fundamental principle of this technique is that ferromagnetic iron particles (Partrac, UK) were spread on the biofilm surface and afterwards attracted by an electromagnetic inductor positioned in a defined distance (4 mm) to the biofilm surface (see a schematic image below). The adhesion force of the biofilm surface is regarded as equivalent to the strength of the magnetic field required to retrieve these ferromagnetic particles and can be deduced from the applied electric current (amperage [mA]). Thus, the measured amperage can be used as a reliable first qualitative proxy for the biofilm stability hereafter denoted as “adhesiveness”. The determination of the biofilm adhesiveness described by Larson et al. uses four different thresholds to describe the varying behavior of the ferromagnetic particles during increasing current [23]. In this study a threshold was chosen which represents the level of amperage where half of the particles are attracted. Furthermore, the gradient of the magnetic field [mT mm-1] could be derived from the amperage after calibration of the electromagnet with a magnetometer (PCE-G28, PCE Instruments, Germany), and is able to describe the actual characteristics of the magnetic field that is responsible for the particle attraction. Thus, results of the determination of biofilm adhesiveness are sown as current [mA] as well as gradient of the magnetic field [mT mm-1] (Table 2 and Figure 1).
| Sample |
Current [mA] |
Gradient [mT mm-1] |
| C |
227.4 ± 24.0 |
0.9 ± 0.3 |
| T1 |
194.2 ± 17.1 |
0.5 ± 0.2 |
| T2 |
186.9 ± 24.5 |
0.4 ± 0.3 |
| T3 |
184.5 ± 14.6 |
0.4 ± 0.2 |
| T4 |
187.8 ± 15.8 |
0.5 ± 0.2 |
Table 2 Biofilm adhesiveness displayed as measured current [mA] and derived gradient of the magnetic field [mT mm-1].
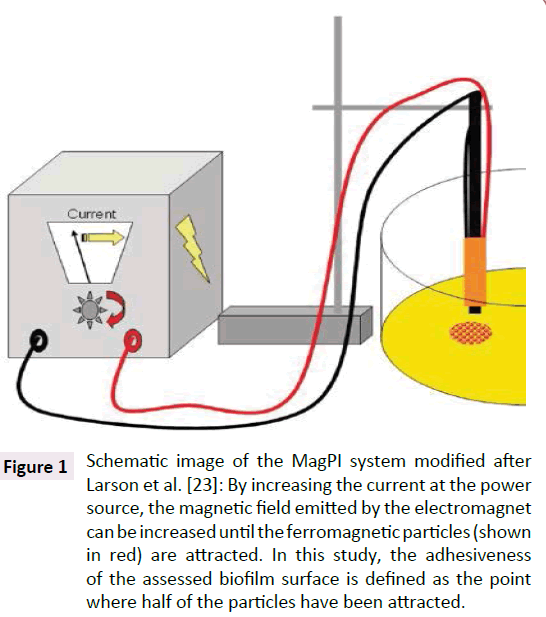
Figure 1:Schematic image of the MagPI system modified after Larson et al. [23]: By increasing the current at the power source, the magnetic field emitted by the electromagnet can be increased until the ferromagnetic particles (shown in red) are attracted. In this study, the adhesiveness of the assessed biofilm surface is defined as the point where half of the particles have been attracted.
Optimization of the MagPI system
As mentioned above, a broad range of modifications of the original MagPI system as described by Larson et al. was performed in order to increase the sensitivity and reproducibility of the gathered results [23]. One major modification was the use of a customized electromagnet with a highly magnetisable μ metal core that displayed exceptionally lower remanence. High remanence as found in different metals may lead to residual magnetism in the electromagnet when the current is already turned off and might thus bias the accuracy of subsequent MagPI measurements. In addition, to ensure the measurement of biofilm adhesiveness on a comparable surface area, the size distribution of the provided ferromagnetic particles was optimized by sequentially sieving resulting in particle diameters between 200 and 350 μm. This facilitated the application of a most comparable number of particles by transferring a defined volume of the particles, and thus ensuring a highly reproducible coated area. Finally, the reproducibility of the MagPI device could be improved by the implementation of an open source microcontroller unit (Arduino Board Model Leonardo R3, Conrad, Germany) allowing the precise trigger of the electromagnet in a series of gradually increasing steps lasting 6 s each [26,27]. In the present study, range finding experiments were performed prior to the actual experiments in order to ensure the most appropriate sensitivity of the MagPI system. Whereas natural freshwater biofilms were shown to potentially possess a very high adhesiveness, the preliminary assessment in this study showed that the value of adhesiveness of mono-species bacterial biofilms can be up to one order of magnitude lower [12]. To further validate the trigger levels given by the microcontroller unit, a high-precision ammeter (Voltcraft VC 950, Conrad, Germany) was integrated enabling the continuous control of the actual electric current.
EPS compounds and structure of the biofilms
Prior to EPS extraction, biofilms were sampled from the glass slide surface. In total, the complete surface of nine slides of each treatment and 18 of the control originated from different petri dishes (nine slides during Phase 1, and nine slides during Phase 2, Table 1) were carefully scraped off using a sterile spatula. The colloidal (water-extractable) EPS compounds were extracted from biofilm samples as described by Gerbersdorf et al. [28]. Subsequently, the contents of EPS carbohydrates and protein were determined using phenol assays and a modified Lowry protocol, respectively [29-31]. To assess the biofilm structure, six biofilm covered glass slides of each treatment, and twelve of the control (six slides during phase 1, and six slides during phase 2, Table 1) were stained with DAPI (final concentration 5 μg l-1) for 15 min, washed with deionized water for 10 min and were finally examined under 418 nm excitation wavelength using an Axioscop fluorescence microscope (Carl Zeiss, Oberkochen, Germany). Images were taken using an Axiocam MRc 5 (Carl Zeiss, Oberkochen, Germany) and image processing was performed using ImageJ (version 1.6.0_24) to calculate the mean rate of covered surface by the bacterial cells [32].
Statistical Analyse
The datasets of biofilm adhesiveness, EPS contents and surface coverage were checked for normal distribution using a Shapiro- Wilk test (confidence interval 95%). Homoscedasticity of data originated from different groups/treatments was evaluated with a Brown-Frosythe test (confidence interval 95%). One-way ANOVA (confidence interval 95%, chi-square approximation, Tukey error protection) was performed for comparison of normally distributed datasets. As only the dataset for surface coverage displayed normal distribution, the Kruskal-Wallis test (with chisquare approximation and Bonferroni error protection) was used to compare the different treatments for biofilm adhesiveness and EPS contents: Mean adhesiveness was compared between the control and the treatments. Subsequently, each treatment was compared with the control and the treatments were compared pairwise: As T1 and T2 were considered as non-independent samples, the comparison of these two treatments used a Friedman test (with Bonferroni error protection), while all other comparisons (C-T1, C-T2, C-T3, C-T4, T1-T3, T1-T4, T2-T3, T3-T4) used a Kruskal-Wallis test (with chi-square approximation and Bonferroni error protection). Mean surface coverage by biofilm cells was compared between control and the treatment with 600 μgl-1 AgNPs (T1) by means of a one-way ANOVA. In addition, mean EPS carbohydrate as well as protein contents were pairwise compared between the control and T1. Correlation between biofilm adhesiveness and EPS contents was tested by calculating Spearman's rank correlation coefficients after pooling control and T1 datasets (correlation between all adhesiveness and EPS carbohydrate values of the control and T1 were evaluated; in a separate approach, correlations between all adhesiveness and EPS protein values of the control and T1 were checked).
Results
Characterization of AgNPs
The mean hydrodynamic diameter (z-average) of citratecoated AgNPs was 34.5 ± 1.2 nm as determined by means of DLS measurements. High polydispersity index of 0.37 ± 0.03 indicated a broad particle size distribution. The major fraction (80%) of AgNPs used in this study was spherical. Residual fraction contained predominantly rod shaped particles and only small amount of triangles. Further detailed information on the particle size distribution and shape of the AgNPs are given in Metreveli et al. [33,34]. The citrate-coated Ag NPs were negatively charged displaying a zeta potential of -59 ± 2 mV that underlines the high stability of the AgNPs in the original dispersions (Figure 2).
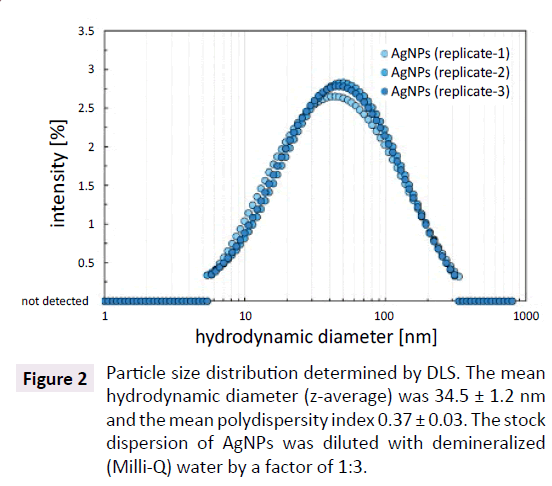
Figure 2:Particle size distribution determined by DLS. The mean hydrodynamic diameter (z-average) was 34.5 ± 1.2 nm and the mean polydispersity index 0.37 ± 0.03. The stock dispersion of AgNPs was diluted with demineralized (Milli-Q) water by a factor of 1:3.
Effects of AgNPs on structural biofilm parameters
Biofilm adhesiveness
The exposure to the different antimicrobial agents (T1-T4) resulted in a significant decrease in biofilm adhesiveness. The mean adhesiveness of the untreated biofilms was 71.5 ± 24.1 mA, while the mean value for biofilms after exposure to 600 μg l-1 AgNPs (T1) was 38.3 ± 17.1 mA. The positive controls treated with 2.4 mg l-1 AgNPs (T2), 2.4 mg l-1 AgNO3 (T3) or 125 mg l-1 CuSO4 (T4) displayed very similar mean adhesiveness as T1 (T2: 31.5 ± 23.8 mA, T3: 28.8 ± 14.4 mA; T4: 32.1 ± 15.7 mA). This observed difference between the untreated control and each treatment was highly significant (Kruskal-Wallis, p<0.0001, n=25). This result is of particular interest, because 600 μg l-1 is close to the predicted environmentally relevant concentration range of AgNPs causing sub-lethal impact reported during previous experiments [35,36]. In addition, the concentrations applied in positive controls (2.4 mg l-1 AgNPs, 2.4 mg l-1 AgNO3, 125 mg l-1 CuSO4) were reported to have a lethal effect on different types of cells and biofilms [37-39]. As no significant difference in biofilm adhesiveness between the four different treatments could be detected, the effect of AgNPs was apparently concentration independent. Thus, the following presentation of the results will focus on the comparison between the untreated control and the biofilm treated with the concentration of 600μg l-1 AgNPs (T1) (Table 2 and Figure 3).
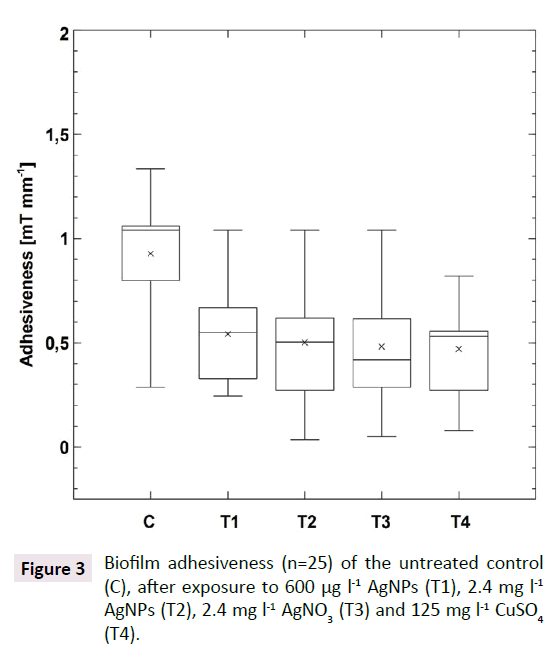
Figure 3: Biofilm adhesiveness (n=25) of the untreated control (C), after exposure to 600 µg l-1 AgNPs (T1), 2.4 mg l-1 AgNPs (T2), 2.4 mg l-1 AgNO3 (T3) and 125 mg l-1 CuSO4 (T4).
EPS compounds and structure of the biofilms
The performed microscopic characterization and image processing displayed major structural differences between exposed and untreated biofilms which demonstrated the response of the biofilm structure to the exposure of AgNPs: In the presence of AgNPs, cells of the homogeneous plain biofilm cluster changed into EPS coated bulky aggregates and thereby leaving a significant fraction of the surface as virtually cell free interspaces. In doing so, the mean surface coverage of 70.3 ± 11.5% of the untreated biofilm was reduced to 41.6 ± 19.1% after the exposure to 600μg l-1 AgNPs which indicated a visible statistical trend (ANOVA, p=0.049, n=12). This observation may also explain the differences in the mean adhesiveness. While the surface of the untreated control was covered to a higher extend with bacterial cells embedded in sticky EPS, the biofilms in T1 displayed large interspaces free of bacterial cells within the biofilm. These areas with low adhesion reduced the measured overall mean adhesiveness of the corresponding surface patches (Figure 4).
The contents of colloidal EPS compounds displayed clear differences between the untreated control and biofilms after the exposure to AgNPs (600 μg l-1): While the mean colloidal content of EPS carbohydrates in the untreated control was 4.67 ± 2.13 μg l-1, the treated biofilms (T1) displayed a mean value of 2.37 ± 0.79 μg l-1. Parallel to this, the mean colloidal EPS protein content determined in the untreated control was 4.56 ± 1.93 μg l-1, whereas the treated biofilms (T1) displayed a mean value of 2.56 ± 0.32 μg l-1. In both cases a significant difference could be shown between the untreated control and T1 (Kruskal-Wallis, for carbohydrates p=0.0020, for proteins p=0.0040, for both n=18). As a result, EPS contents and biofilm adhesiveness displayed high correlations (Spearman, for carbohydrates-adhesiveness p=0.0027, r=0.4900, for proteins-adhesiveness p=0.0166, r=0.4000) (Figure 5).
Discussion
Among other engineered inorganic nanoparticles, AgNPs are most widely used in two different ways: either as an addition to various products, e.g. textiles, cosmetics and pharmaceutics [40] or as coatings, e.g. in implants [41,42]. Xiu et al. [43] suggested that the antimicrobial activity of AgNPs is caused by the release of Ag+ ions and the destabilizing effect of Ag+ ions on the biofilm matrix could be demonstrated by Chaw et al. [44]. However, the characterization of the AgNPs utilized in this study demonstrated only a very small release of the dissolved silver from nanoparticles. As observed in our earlier study 34, one day after dispersion of the similarly synthesized citratecoated AgNPs in the R2A medium, only 0.1% of the initially spiked 2 mg l−1 total Ag was released in the solution, resulting in a concentration of 3.9 ± 2.2 μg l−1. According to the species calculation, the released silver was predominantly present as Ag+ ions (99.4%) [34]. Nevertheless, a clear effect of the treatment with AgNPs could be detected regarding biofilm EPS contents, biofilm structure and adhesiveness. Thus, it can be assumed that the effect of silver nanoparticles upon biofilm functionality and organisation was not exclusively due to the release of silver ions. Although current studies suggest the release of radical anions in the presence of AgNPs which potentially damage cell membranes, the exact mechanisms of operation of AgNPs is still not fully unraveled [45,46]. Furthermore, the colloidal stability of the nanoparticles in the biological test media should be also considered. As observed in our earlier study, the zeta potential of similar citrate-coated AgNPs after dispersion in R2A medium was less negative (-27 ± 1 mV) than in original dispersion (-59 ± 2 mV) due to the compression of electrical double layer at high ionic strength of the medium (8.75 mmol l-1) [34]. Despite this, as shown in our earlier study, dispersion of AgNPs in the R2A medium leads only to limited aggregation [34]. The z-average hydrodynamic diameter of AgNPs increases rapidly only within the first few seconds and then remains constant at ca. 65 nm. The R2A medium contains surfactant Tween 80 which is able to stabilize nanoparticles [47]. The R2A medium contains also proteins and protein fragments which can also interact with nanoparticles, form a protein corona and affect their stability. The formation of protein corona on the surface of AgNPs may protect the nanoparticles against dissolution [48].
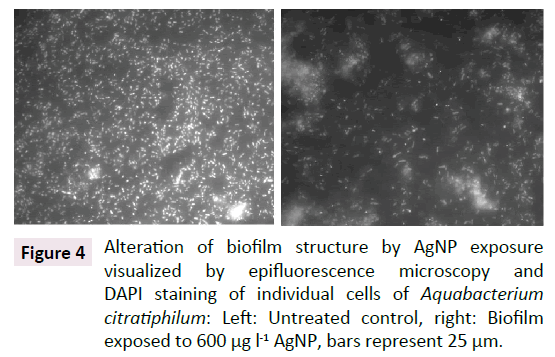
Figure 4: Alteration of biofilm structure by AgNP exposure visualized by epifluorescence microscopy and DAPI staining of individual cells of Aquabacterium citratiphilum: Left: Untreated control, right: Biofilm exposed to 600 µg l-1 AgNP, bars represent 25 µm.
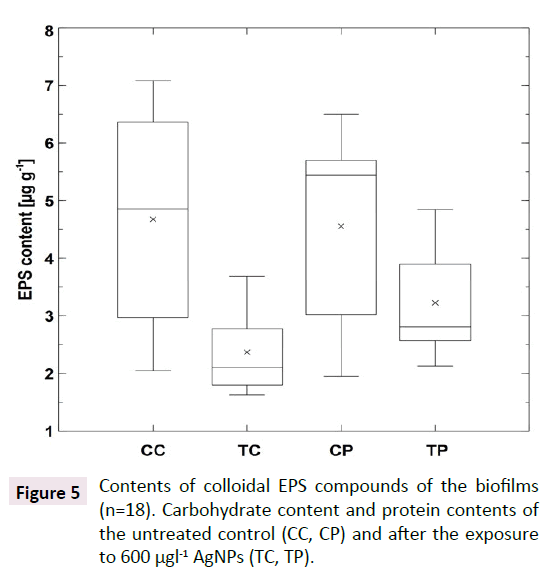
Figure 5: Contents of colloidal EPS compounds of the biofilms (n=18). Carbohydrate content and protein contents of the untreated control (CC, CP) and after the exposure to 600 µgl-1 AgNPs (TC, TP).
In addition, up to now there is no clear and conclusive picture about the effects of a treatment with AgNPs upon different biofilms and results of some studies are even contradictive. On the one hand, different studies described the antibacterial effect of AgNPs especially on mono-species biofilms of clinical relevant bacteria and there are indications that other cells and organisms can also be impacted [49-53]. Furthermore, the possibility of AgNP treatment to affect the community composition of beneficial biofilms e.g. in activated sludge of wastewater treatment plants was described [54,55]. On the other hand, Kroll et al. [16] stated that in contrast to Ag+ ions, AgNPs apparently have no direct impacts on biomass or diversity, but on EPS production and the three dimensional structure of benthic biofilms [16]. In general, the formation of a mucilage layer composed of EPS constitutes an essential step during the development of a biofilm and the EPS matrix has various different ecological functions including the provision of a nutrient reservoir and the formation of a protective layer against desiccation and potentially harmful substances [56]. Furthermore, the importance of EPS proteins and carbohydrates for the bacterial attachment to different surfaces was repeatedly reported [57-59]. Further studies emphasized that extracellular carbohydrates constitute a driving factor for biofilm formation and stability [60-62]. This reported high correlation between biofilm stability and EPS carbohydrate content was recently confirmed by Schmidt et al. [12]. However, in the mentioned study the stability of the biofilm was demonstrated to be dependent on extracellular proteins, too. These findings are in good correlation to the results reported here: a significant decrease in the content of EPS compounds is directly proportionate to a highly significant reduction in the adhesiveness of the biofilm. This gives first insight into the highly complex effects caused by an exposure to sub-lethal concentrations of AgNPs. Even if an exposure to 600 μg l-1 AgNPs can be assumed as non-lethal for individual biofilm cells, their cellular metabolism and the resulting functionality of the EPS matrix may be significantly affected, which influences the overall biofilm morphology and stability. These observations and results of previous experiments with mono-species biofilms of Aquabacterium citratiphilum may be correlated to a process in biofilms described by Kang et al. [63] where Ag+ ions are detoxified by reduction to AgNPs via the EPS matrix which apparently grants a high capability to detoxify silver to natural phototrophic biofilms as suggested by Gonzalez et al. [18]. In addition, Choi et al. [64] could demonstrate that planktonic bacterial cells can be much more susceptible to AgNPs than matured biofilms of the same species of bacteria suggesting the protective role of the extracellular mucilage.
As stated above, the AgNPs concentration used in the present study may be considered as non-lethal due to results of previous experiments [19]. However, the effects on biofilm adhesiveness are comparable to the reported decrease in biofilm stability after a treatment with Triclosan which is another widely used antibiotic even when low concentrations of the antibiotic were used [14]. This is of particular interest, as this particular investigation is one of just a few assessments of the impact of anthropogenic influences on microbial biostabilization. Results of this mentioned study suggest a correlation between bacterial biomass as well as the diversity of the bacterial community and the stability of the biofilm system. However, the differences in EPS contents between untreated control and the different treatments appeared less distinct than in the present investigation. A possible explanation may be found in the fact that in contrast to our study examining mono-species Aquabacterium biofilms, natural complex bacterial assemblages were used. Thus, functional redundancies within different species of the natural bacterial community appear plausible that were not present in the Aquabacterium biofilms and resulted in a clearer decrease in EPS contents. Nevertheless, results suggest that even if the mode of operation differs between non-lethal AgNPs and antibacterial Triclosan, the effects on the overall functionality of biofilm system can be very similar.
These various findings demonstrate the complexity of the various possible effects of a treatment or coating of a surface with AgNPs. However, it is important to note that higher concentrations of AgNPs-compared to the used concentrations in our study-could be demonstrated to impact different non-target organisms [65-68]. As prolonged accumulation of nanoparticles in the environment may result in these higher concentrations in the future, methods to determine AgNPs concentration in environmental samples have been developed and optimized in the last years [69]. This optimizations of analytics allow insights in the current status quo e.g. if accumulation in the biosphere occurs and if this can be linked to adverse effects on a range of different organisms as well as ecosystem services including microbial biostabilization of contaminated fine lotic sediments. Thus, it is essential to possess suitable analytic tools to monitor the biofilm and sediment stability of affected sites in the environment. The presented optimized MagPI system constitutes a cost-effective, fast and reliable method to determine the adhesiveness of surfaces and its non-destructive measurement principle facilitates data generation in very high temporal and spatial resolution. Due to the high sensitivity, it can be seen as an essential link between biological and engineering, industrial or medical approaches e.g. aiming to incorporate the impact of biofilms in sediment dynamics or investigating the effects of various substances on biofilm stability, adhesives and the resulting ecosystem functions such as microbial biostabilization [70].
Conclusion
This study could provide essential insights into the impact of an exposure of biofilms to a low-dose, sub-lethal, environmentally and ecologically relevant concentration of AgNPs: Although previous studies strongly suggested no lethal impact on the level of individual bacterial cells in the biofilm system, a clear structural and functional response of the biofilm network became clear: The production and composition of biofilm EPS, the structure of the biofilm as well as the overall biofilm adhesiveness was significantly impacted in by the presence of AgNPs. Especially the fact that the decrease in biofilm adhesiveness apparently was not dependent on the concentration of this putative antimicrobial agent demonstrated the complexity of the biofilm system and the interaction mechanisms of AgNPs. Thus, this study could demonstrate the importance of a thorough determination of the adhesiveness of a biofilm system to gather further insights into fundamental biofilm processes: While the determined biofilm adhesiveness constitutes an important marker for biofilm stability, more information about the biofilm functionality could be derived from this parameter. Examples are the nutritional or physiological state of the microbes in the biofilm and the capability of the biofilm to attract and bind flocs, particles or pollutants or to colonize and stabilize new surfaces of fine sediments. Thus, the MagPI based determination of biofilm adhesiveness could be an essential tool to perform a more process orientated, systemic analysis of any kind of biofilm formation.
Acknowledgement
This study was kindly supported by the German Research Foundation (DFG, Research unit INTERNANO: FOR 1536, subprojects BIOFILM MA3273/3-2 and MASK SCHA849/16-2).
References
- Yallop ML, Paterson DM, Wellsbury P (2000) Interrelationships between rates of microbial production, exopolymer production, microbial biomass and sediment stability in biofilms of intertidal sediments. Microb Ecol 39: 116-127.
- Gerbersdorf SU, Bittner R, Lubarsky H, Manz W, Paterson DM (2009) Microbial assemblages as ecosystem engineers of sediment stability. J Soils Sediments 9: 640-652.
- Lubarsky HV, Hubas C, Chocholek M, Larson F, Manz W, et al. (2010) The stabilisation potential of individual and mixed assemblages of natural bacteria and microalgae. Plos One 5.
- Karickhoff SW, Brown DS, Scott TA (1979) Sorption of hydrophobic pollutants on natural sediments. Water Res 13: 241-248.
- Mueller-Steinhagen H, Malayeri MR, Watkinson AP (2011) Heat exchanger fouling: Mitigation and cleaning strategies. Heat Transfer Eng 32: 189-196.
- Makino T, Jimi S, Oyama T, Nakano Y, Hamamoto K, et al. (2015) Infection mechanism of biofilm-forming Staphylococcus aureus on indwelling foreign materials in mice. Int Wound J 12: 122-131.
- Nishitani K, Sutipornpalangkul W, Bentley KLDM, Varrone JJ, Bello-Irizarry SN, et al. (2015) Quantifying the natural history of biofilm formation in vivo during the establishment of chronic implant-associated Staphylococcus aureus osteomyelitis in mice to identify critical pathogen and host factors. J Orthopaed Res 33: 1311-1319.
- Horn H, Lackner S (2014) Modeling of biofilm systems: A review. Adv Biochem Eng Biotechnol 146: 53-76.
- Merkey BV, Chopp DL (2014) Modeling the impact of interspecies competition on performance of a microbial fuel cell. Bull Math Biol 76: 1429-1453.
- Miller JK, Brantner JS, Clemons C, Kreider KL, Milsted A, et al. (2014) Mathematical modelling of Pseudomonas aeruginosa biofilm growth and treatment in the cystic fibrosis lung. Math Med Biol 31: 179-204.
- Thom M, Schmidt H, Gerbersdorf SU, Wieprecht S (2015) Seasonal biostabilization and erosion behavior of fluvial biofilms under different hydrodynamic and light conditions. Int J Sediment Res 30: 273-284.
- Schmidt H, Thom M, King L, Wieprecht S, Gerbersdorf SU (2016) The effect of seasonality upon the development of lotic biofilms and microbial biostabilisation. Freshwater Biol.
- Mason CF, Underwood GJC, Baker NR, Davidson I, Hanlon A, et al. (2003) The role of herbicides in the erosion of salt marshes in eastern England. Environ Pollut 122: 41-49.
- Lubarsky HV, Gerbersdorf SU, Hubas C, Behrens S, Ricciardi F, et al. (2012) Impairment of the bacterial biofilm stability by triclosan. Plos One 7.
- Reidy B, Haase A, Luch A, Dawson KA, Lynch I (2013) Mechanisms of silver nanoparticle release, transformation and toxicity: A critical review of current knowledge and recommendations for future studies and applications. Materials 6: 2295-2350.
- Kroll A, Matzke M, Rybicki M, Obert-Rauser P, Burkart C, et al. (2015) Mixed messages from benthic microbial communities exposed to nanoparticulate and ionic silver: 3D structure picks up nano-specific effects, while EPS and traditional endpoints indicate a concentration-dependent impact of silver ions. Environ Sci Pollut Res. 1-17.
- Schaumann GE, Philippe A, Bundschuh M, Metreveli G, Klitzke S, et al. (2015) Understanding the fate and biological effects of Ag and TiO2 nanoparticles in the environment: The quest for advanced analytics and interdisciplinary concepts. Sci Total Environ 535: 3-19.
- Gonzalez AG, Mombo S, Leflaive J, Lamy A, Pokrovsky OS, et al. (2015) Silver nanoparticles impact phototrophic biofilm communities to a considerably higher degree than ionic silver. Environ Sci Pollut Res 22: 8412-8424.
- Grun AY, Meier J, Metreveli G, Schaumann GE, Manz W (2016) Sub lethal concentrations of silver nanoparticles affect the mechanical stability of biofilms. Environ Sci Pollut Res 23: 24277-24288.
- Kalmbach S, Manz W, Wecke J, Szewzyk U (1999) Aquabacterium gen. nov., with description of Aquabacterium citratiphilum sp. nov., Aquabacterium parvum sp. nov. and Aquabacterium commune sp. nov., three in situ dominant bacterial species from the Berlin drinking water system. Int J Syst Bacteriol 49: 769-777.
- Kalmbach S, Manz W, Bendinger B, Szewzyk U (2000) In situ probing reveals Aquabacterium commune as a widespread and highly abundant bacterial species in drinking water biofilms. Water Res 34: 575-581.
- Windler M, Leinweber K, Bartulos CR, Philipp B, Kroth PG (2015) Biofilm and capsule formation of the diatom Achnanthidium minutissimum are affected by a bacterium. J Phycol 51: 343-355.
- Larson F, Lubarsky H, Gerbersdorf SU, Paterson DM (2009) Surface adhesion measurements in aquatic biofilms using magnetic particle induction: MagPI. Limnol Oceanogr. 7: 490-497.
- Metreveli G, Frombold B, Seitz F, Grün A, Philippe A, et al. (2016) Impact of chemical composition of ecotoxicological test media on the stability and aggregation status of silver nanoparticles. Environ Sci Nano 3: 418-433.
- Turkevich J, Stevenson PC, Hillier J. (1951) A study of the nucleation and growth processes in the synthesis of colloidal gold. Discuss of the Faraday Soc 11: 55-75.
- Thom M, Schmidt H, Wieprecht S, Gerbersdorf S (2015) Biostabilization of fluvial sediments: An improved device to address an old problem. IAHR, Den Haag, Netherlands.
- Thom M, Schmidt H, Wieprecht S, Gerbersdorf SU (2016) The role of surface adhesion in biostabilisation processes. Proceedings of the International Symposium on River Sedimentation ISRS, Stuttgart.
- Gerbersdorf SU, Manz W, Paterson DM (2008) The engineering potential of natural benthic bacterial assemblages in terms of the erosion resistance of sediments. Fems Microbiol Ecol 66: 282-294.
- Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28: 350-356.
- Raunkjaer K, Hvitvedjacobsen T, Nielsen PH (1994) Measurement of pools of protein, carbohydrate and lipid in domestic wastewater. Water Res 28: 251-262.
- Frolund B, Palmgren R, Keiding K, Nielsen PH (1996) Extraction of extracellular polymers from activated sludge using a cation exchange resin. Water Res 30: 1749-1758.
- Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9: 671-675.
- Metreveli G, Philippe A, Schaumann GE (2015) Disaggregation of silver nanoparticle homoaggregates in a river water matrix. Sci Total Environ 535: 35-44.
- Metreveli G, Frombold B, Seitz F, Grün A, Philippe A, et al. Impact of chemical composition of ecotoxicological test media on the stability and aggregation status of silver nanoparticles.
- Gottschalk F, Sun T, Nowack B (2013) Environmental concentrations of engineered nanomaterials: Review of modeling and analytical studies. Environ Pollut 181: 287-300.
- Grün A, Meier J, Metrevelli G, Schaumann G, Manz W (2016) Sublethal concentrations of silver nanoparticles affects the mechanical stability of biofilms. Environ Sci Pollut Res 23: 24277–24288.
- Ratte HT (1999) Bioaccumulation and toxicity of silver compounds: A review. Environ Toxicol Chem 18: 89-108.
- Choi O, Deng KK, Kim NJ, Ross L, Surampalli RY, et al. (2008) The inhibitory effects of silver nanoparticles, silver ions and silver chloride colloids on microbial growth. Water Res 42: 3066-3074.
- Baker J, Sitthisak S, Sengupta M, Johnson M, Jayaswal RK, (2010) Copper stress induces a global stress response in Staphylococcus aureus and represses sae and agr expression and biofilm formation. Appl Environ Microbiol 76: 150-160.
- Ahamed M, AlSalhi MS, Siddiqui MKJ (2010) Silver nanoparticle applications and human health. Clinica Chimica Acta 411: 1841-1848.
- Monteiro DR, Gorup LF, Takamiya AS, Ruvollo-Filho AC, Camargo ER, et al. (2009) The growing importance of materials that prevent microbial adhesion: Antimicrobial effect of medical devices containing silver. Int J Antimicrob Ag 34: 103-110.
- Knetsch MLW, Koole LH (2011) New strategies in the development of antimicrobial coatings: The example of increasing usage of silver and silver nanoparticles. Polymers. 3: 340-366.
- Xiu Z, Zhang Q, Puppala HL, Colvin VL, Alvarez PJJ (2012) Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Lett 12: 4271-4275.
- Chaw KC, Manimaran M, Tay FEH (2005) Role of silver ions in destabilization of intermolecular adhesion forces measured by atomic force microscopy in Staphylococcus epidermidis biofilms. Antimicrob Agents Ch 49: 4853-4859.
- Tripathi GNR (2003) p-Benzosemiquinone radical anion on silver nanoparticles in water. J Am Chem Soc 125: 1178-1179.
- Hussain SM, Hess KL, Gearhart JM, Geiss KT, Schlager JJ (2005) In vitro toxicity of nanoparticles in BRL 3A rat liver cells. Toxicol In Vitro 19: 975-983.
- Li X, Lenhart JJ (2012) Aggregation and dissolution of silver nanoparticles in natural surface water. Environ Sci Technol 46: 5378-5386.
- Kakinen A, Ding F, Chen PY, Mortimer M, Kahru A, et al. (2013) Interaction of firefly luciferase and silver nanoparticles and its impact on enzyme activity. Nanotechnology 24: 9.
- Sotiriou GA, Pratsinis SE (2010) Antibacterial activity of nanosilver ions and particles. Environ Sci Technol 44: 5649-5654.
- Kim JH, Kang EJ, Park MG, Lee BG, Kim KY (2011) Effects of temperature and irradiance on photosynthesis and growth of a green-tide-forming species (Ulva linza) in the Yellow Sea. J Appl Phycol 23: 421-432.
- Morones JR, Elechiguerra JL, Camacho A, Holt K, Kouri JB, et al. (2005) The bactericidal effect of silver nanoparticles. Nanotechnology. 16: 2346-2353.
- Miura N, Shinohara Y (2009) Cytotoxic effect and apoptosis induction by silver nanoparticles in HeLa cells. Biochem Bioph Res Co 390: 733-737.
- Yeo MK, Yoon JW (2009) Comparison of the effects of nano-silver antibacterial coatings and silver ions on zebra fish embryogenesis. Mol Cell Toxicol 5: 23-31.
- Sun X, Sheng Z, Liu Y (2013) Effects of silver nanoparticles on microbial community structure in activated sludge. Sci Total Environ 443: 828-835.
- Shang ZY, Nostrand JDV, Zhou JZ, Liu Y (2015) The effects of silver nanoparticles on intact wastewater biofilms. Front Microbiol 6: 11.
- Flemming HC, Wingender J (2010) The biofilm matrix. Nature R Microbiol 8: 623-633.
- Krogfelt KA, Bergmans H, Klemm P (1990) Direct evidence that the FIMH protein is the mannose-specific adhesin of Escherichia coli type 1 fimbriae. Infect Immun 58: 1995-1998.
- Nallapareddy SR, Qin X, Weinstock GM, Hook M, Murray BE (2000) Enterococcus faecalis adhesin, ace, mediates attachment to extracellular matrix proteins collagen type IV and laminin as well as collagen type I. Infect Immun 68: 5218-5224.
- Li GL, Smith CS, Brun YV, Tang JX (2005) The elastic properties of the Caulobacter crescentus adhesive holdfast are dependent on oligomers of N-acetylglucosamine. J Bacteriol 187: 257-265.
- Sutherland TF, Grant J, Amos CL (1998) The effect of carbohydrate production by the diatom Nitzschia curvilineata on the erodibility of sediment. Limnol Oceanogr 43: 65-72.
- Underwood GJC, Paterson DM (2003) The importance of extracellular carbohydrate production by marine epipelic diatoms. Adv Bot Res 40: 183-240.
- Tolhurst TJ, Defew EC, de-Brouwer JFC, Wolfstein K, Stal LJ, et al. (2006) Small-scale temporal and spatial variability in the erosion threshold and properties of cohesive intertidal sediments. Cont Shelf Res 26: 351-362.
- Kang F, Alvarez PJ, Zhu D (2014) Microbial extracellular polymeric substances reduce Ag+ to silver nanoparticles and antagonize bactericidal activity. Environ Sci Technol 48: 316-322.
- Choi O, Yu CP, Fernandez GE, Hu Z (2010) Interactions of nanosilver with Escherichia coli cells in planktonic and biofilm cultures. Water Res 44: 6095-6103.
- Chae YJ, Pham CH, Lee J, Bae E, Yi J, et al. (2009) Evaluation of the toxic impact of silver nanoparticles on Japanese medaka (Oryzias latipes). Aquat Toxicol 94: 320-327.
- Farkas J, Christian P, Urrea JAG, Roos N, Hassellöv M, et al. Effects of silver and gold nanoparticles on rainbow trout (Oncorhynchus mykiss) hepatocytes. Aquat Toxicol 96: 44-52.
- Laban G, Nies LF, Turco RF, Bickham JW, Sepulveda MS (2010) The effects of silver nanoparticles on fathead minnow (Pimephales promelas) embryos. Ecotoxicology 19: 185-195.
- Yin L, Colman BP, McGill BM, Wright JP, Bernhardt ES (2012) Effects of silver nanoparticle exposure on germination and early growth of eleven wetland plants. Plos One: 7.
- Zook JM, Long SE, Cleveland D, Geronimo CLA, MacCuspie RI (2011) Measuring silver nanoparticle dissolution in complex biological and environmental matrices using UV-visible absorbance. Anal Bioanal Chem 401: 1993-2002.
- Righetti M, Lucarelli C (2007) May the Shields theory is extended to cohesive and adhesive benthic sediments? J Geophys Res, p: 112.