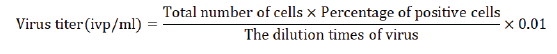Keywords
Cytometry; Baculovirus; Cells; Titer
Introduction
Baculovirus expression vector system Baculovirus Expression
Vector System (BEVS) can express foreign genes at a high level.
The biological characteristics of the expression product are similar
to those of natural products. It has good application prospects in
vaccine and drug development. Seven products including cancer
vaccines and influenza vaccines have been approved, including
vaccines and therapeutic products [1]. It is worth mentioning that
on November 16, 2020, the new coronavirus recombinant protein
vaccine developed by West China Hospital of Sichuan University
using insect cells has entered the phase II clinical stage [2].
The determination of baculovirus titer is very important for the
control and optimization of recombinant protein expression
process parameters. Baculovirus determination methods mainly
include plaque method, endpoint dilution method Terminology
Median Tissue Culture Infectious Dose (TCID50), live cell size
determination method, fluorescent quantitative Polymerase
Chain Reaction (PCR) method, flow cytometry detection method,
immunostaining method, colorimetric indicator, and β-half
determination. Lactosidase activity, the use of microfluidic
bioanalyzer, etc [3]. The plaque method is a classic method for
titer determination, but the detection cycle is long (6-10 days),
and the operator's technical and experience requirements are
high the end-point dilution method also requires at least 3 days.
Currently, immunostaining and Quantitative Polymerase Chain
Reaction (QPCR) are widely used. The Quantitative Polymerase
Chain Reaction (QPCR) method has the advantage of a short
detection cycle, but it requires a nucleic acid extraction step,
which is to detect the number of gene copies, rather than the
infectious virus titer value, which is not accurate enough. The
immunostaining method also has a series of shortcomings: there
are many operating steps and a long detection cycle; manual
counting is required, which is more labor-intensive; insect cells
are semi-adherent cells, which are not firmly attached during
detection, and they may fall off in the multi-well plate. In the
process of counting spots, the subjectivity is strong and requires
certain experience or standards to distinguish.
The Futures Commission Merchant (FCM) method is a technique
commonly used to identify and isolate specific cells from mixed
samples [4]. It can be used to detect viruses and cells infected
by viruses [5]. Gp64 protein locates in the cytoplasm in the early
stage of virus infection, then migrates to the plasma membrane,
and can be expressed on the surface of infected insect cells
within 6 hours of infection [6,7]. Refer to the relevant literature,
combined with the actual application, stain the cells with
fluorescently labeled anti-gp64-APC antibody, and establish an
Futures Commission Merchant (FCM) method for rapid detection
of baculovirus titer and may affect the titer detection The value
of incubation time, cell state, antibody concentration and other
factors have been optimized and verified [8].
Materials and Methods
Viruses and cells
Recombinant H5N1-HANA、M1、M2、NP Baculovirus,
Recombinant H1N1-HA Baculovirus, Recombinant HEV
Baculovirus, Recombinant 2019-nCOV-N Baculovirus,
2019-nCOV-S Baculovirus, 2019 -nCOV-S1、RBD、RBD dimer
baculovirus and empty baculovirus were prepared by the No.7
Research Laboratory, National Vaccine and Serum Institute; Sf9
insect cells were purchased from ATCC; ExpiSf9 insect cells were
purchased from Thermo Fisher Scientific.
Reagents and instruments
SFX-Insect medium was purchased from GE Baculovirus Envelope
gp64 Monoclonal Antibody (AcV1), APC, and eBioscience™
were purchased from ThermoFisher Scientific; TC Plate 24 Well,
Suspension, F were purchased from Sarstedt; Gibco fetal bovine
serum was purchased from Thermo Scientific PBS and resuspension
(including 2% FBS), self-prepared. FACSCaliburTM flow cytometer
and Falcon flow cytometry tube were purchased from BD; 3-18K
refrigerated centrifuges was purchased from Sigma; QT-2 vortex
mixer was purchased from Shanghai Qite Analytical Instrument
Co., Ltd.; ZWY -211C constant temperature culture oscillator
was purchased from Shanghai Zhicheng Analytical Instrument
Manufacturing Co., Ltd.; Countstat® BioTech automatic cell
counter was purchased from Shanghai Ruiyu Biotechnology Co.,
Ltd.
Method establishment
Add 1 ml logarithmically diluted HEV virus solution and 800 μl to a
24-well suspension culture plate with a density of 1.25 × 10 [6] cells
logarithmic phase insect cells, set up multiple wells and negative
control wells. 27°C shaker 225 rpm, 27°C suspension culture for 14-
16 hours, aspirate the liquid from each well to the flow cytometry
tube and centrifuge to pellet the cells (300 × g, 5 minutes), discard
the supernatant, and add 100 μl at a concentration of 0.15 μg/
ml Stain the cells with the gp64-APC antibody, vortex and mix for
3 to 5 seconds, let stand at room temperature for 30 minutes,
centrifuge to discard the supernatant, slowly add 1 ml PBS along
the tube wall to wash, add 1 ml resuspension solution to resuspend
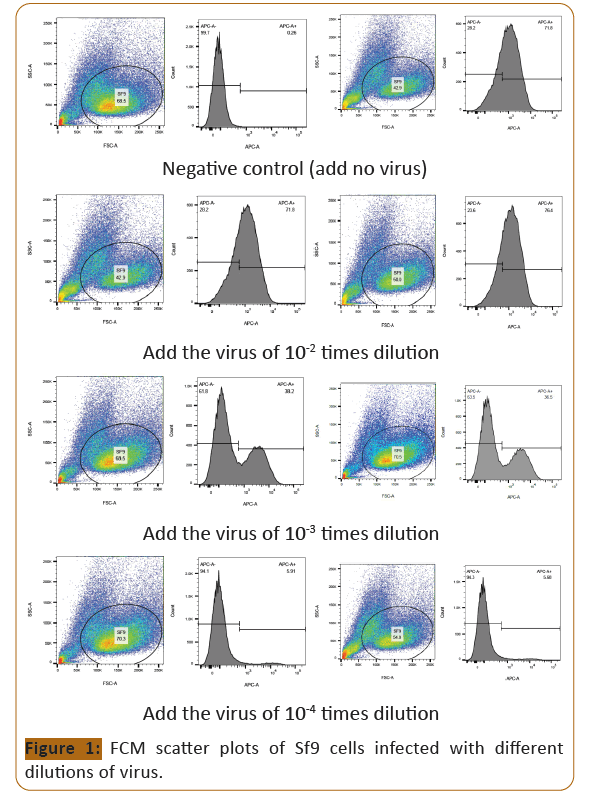
the cells (Figure 1). Flow cytometry detection; select the detection
value of positive cells less than 10% to calculate the virus titer.
Figure 1:FCM scatter plots of Sf9 cells infected with different
dilutions of virus.
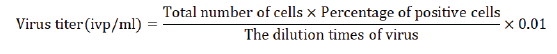
Calculation formula (Infectious virus particle, ivp: infectious virus
particle):
Virus titer(ivp/ml)=(Total number of cells×Percentage of positive
cells)/(The dilution times of virus)×0.01
Method optimization
Incubation time: After 6-48 hours of baculovirus inoculation and
culture, test to observe cell growth and titer change trends to
determine the best culture time.
Concentration of gp64 antibody: Add 100 μl of gp64-APC antibody
with concentrations of 0.05, 0.1, 0.15, 0.2, 0.4 μg/ml for detection.
Observe the trend of titer changes and select the best antibody
concentration.
Concentration of inactivated FBS in dilute diluent: Use HEV
virus with 0%, 0.2%, 0.5%, 0.8%, 1%, 2%, 5%, 8%, 10% dilutions
to resuspend stained cells washed with PBS and observe different
Fasting blood sugar (FBS) contents Whether the resuspension of
the solution affects the detection value of titer, and choose the
best Fasting blood sugar (FBS) concentration.
Cell growth status: Use cells of different growth periods (early,
middle, late, plateau, and decay periods of logarithmic growth)
and different cell generations for titer detection.
1.4.4.2 Use another recombinant baculovirus: 2019-nCOV-RBD
dimer-working virus library to verify the influence of cell growth
status on the detection value.
Method verification
Versatility: Use the Futures Commission Merchant (FCM)
method to detect baculoviruses carrying different foreign genes
(H5N1-HA, H5N1-NA, H5N1-M1, H5N1-M2, H5N1-NP, H1N1-
HA, 2019-nCOV-N, 2019-nCOV-S) Perform testing to verify the versatility of the method.
Repeatability: For the same virus sample, repeat the test 6 times and calculate the Relative standard deviation (RSD) value.
Intermediate precision: The same sample was repeatedly tested by three different operators to calculate the RSD value.
Method application: The Futures Commission Merchant (FCM) method was used to detect the titer of 2019-nCOV-S1, 2019-nCOV-
RBD, and 2019-nCOV-RBD dimer in the tertiary virus seed library. The results were compared with the detect value of the immune-
fluorescence method.
Results
Method establishment
Use the P44 generation, 1 day after the passage of Sf9 cells, to
detect the HEV virus, the detection values were 5.91 × 108 ivp/
ml, 5.68 × 108 ivp/ml , the average value was 5.79 × 108 ivp/ml.
Method optimization
Incubation time: The cell and virus culture time is less than 13
hours, and the detection value is seriously low; between 13~22
hours, the detection value is not much different; between 26~32
hours, the detection value is reduced; between 36~40 hours,
the detection value has a slight upward trend; At 42 hours, the
detection signal decreased significantly and almost disappeared;
between 44~48 hours, the detection value increased significantly
again (Figure 2).
Figure 2:Changes of virus titer detection values at different
incubation times.
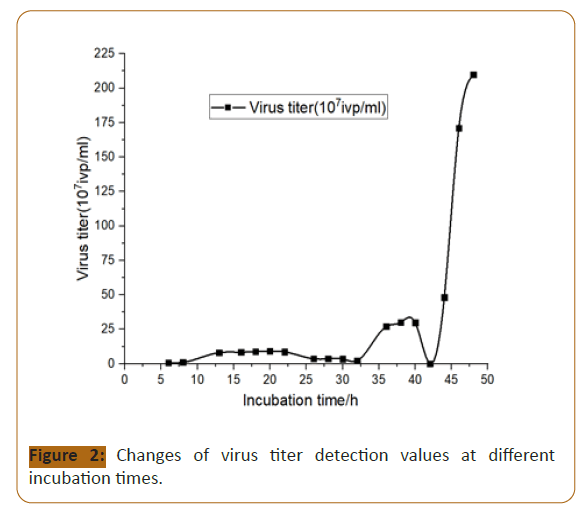
As the culture time increases, the cells that are not infected by
the virus will continue to expand and the cell density will increase
(Figure 3).
Figure 3:Changes of the total number of living cells infected with
different culture time and different dilution ratio of virus.
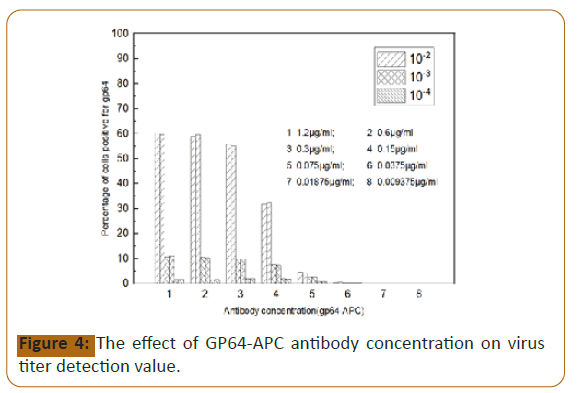
GP64-APC antibody concentration: The concentration of 100 μl
gp64-APC antibody for cell staining between 0.15~1.2 μg/ml
has little effect on the detection value, and has no effect on the
detection value of about 10% positive cell wells, so 0.15 μg/ml is
selected as the detection antibody concentration (Figure 4). If it
is less than 0.15 μg/ml, it will seriously affect the detection value;
if it is less than 0.0375 μg/ml, the proportion of positive cells in
each dilution cannot be distinguished from the negative control
(Figure 5).
Figure 4:The effect of GP64-APC antibody concentration on virus
titer detection value.
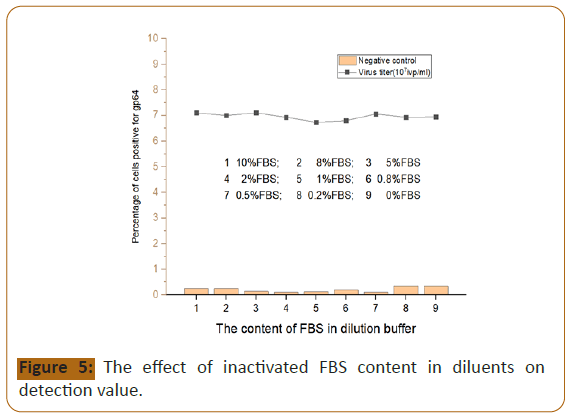
Figure 5:The effect of inactivated FBS content in diluents on
detection value.
The content of inactivated FBS in the diluent: The content
of inactivated Fasting blood sugar (FBS) in the dilution has no
significant effect on the detection value of HEV virus titer. The
average detection value of 9 groups is 6.95, and the proportion
of gp64 positive cells in the 2% Fasting blood sugar (FBS) group is
6.93, which is close to the mean value; the negative control value
between each group (Detection background) The difference is not
obvious, the 2% concentration group is 0.11, which is relatively
low. Therefore, the content of inactivated Fasting blood sugar
(FBS) in the diluent is 2%.
Cell growth status: According to the detection value of Sf9 cell
titer of different cell cycle stages it can be seen that the detection
value of the cells in the early stage of logarithmic growth is
higher and the stability is better (Table 1). Use the cells in the
adaptive and plateau phase, the detection value is lower; The
cells in the recession cannot be used for titer detection due to
the extremely poor cell state or low viability; The cell generation
had no significant effect on the detection value of virus titer.
High-passage cells (119 generations) are not suitable for titer
detection; but, after 3 passages (122 generations), they can be
used for titer detection again (Table 2).
| Generation |
Culture days |
Viable cell density(106cells/ml) |
Viability (%) |
Diameter (μm) |
Phase |
Titer-1 (107ivp/ml) |
Titer-2 (107ivp/ml) |
Titer-average (107ivp/ml) |
| 40 |
2 |
2.97 |
97.51 |
16.98 |
Early-logarithmic |
9 |
8.8 |
8.9 |
| 40 |
5 |
11.2 |
97.35 |
17.67 |
plateau |
4.9 |
4.7 |
4.8 |
| 42 |
3 |
3.6 |
96.4 |
16.7 |
Early-logarithmic |
8.3 |
8.1 |
8.2 |
| 44 |
1 |
1.4 |
95.35 |
16.53 |
Early-logarithmic |
8.8 |
9 |
8.9 |
| 44 |
3 |
5.52 |
98.96 |
16.03 |
Middle-logarithmic |
7.1 |
7.1 |
7.1 |
| 47 |
1 |
1.6 |
98.39 |
16.46 |
adaptive |
7.7 |
7.5 |
7.6 |
| 47 |
2 |
3.5 |
98.71 |
16.28 |
Early-logarithmic |
8.1 |
8.3 |
8.2 |
| 48 |
1 |
1.3 |
98.83 |
16.5 |
adaptive |
7.5 |
7.1 |
7.3 |
| 54 |
7 |
3 |
29.8 |
15.55 |
Recession |
Not-suitable |
Not-suitable |
Not-suitable |
| 55 |
5 |
7.41 |
69.84 |
16.34 |
recession |
Not-suitable |
Not-suitable |
Not-suitable |
| 60 |
2 |
3.7 |
98.72 |
16.06 |
Early-logarithmic |
8.4 |
8.2 |
8.3 |
| 119 |
4 |
8.92 |
90.58 |
17.57 |
Later-logarithmic |
Not-suitable |
Not-suitable |
Not-suitable |
| 122 |
1 |
2.78 |
92.5 |
17.02 |
Early-logarithmic |
8 |
8.7 |
8.4 |
Table 1: Comparison of titer values of Sf9 cells in different cell cycle stages.
| Sample |
SF9/APC-A+ | Freq. of parent |
| Sf9-P119-4 |
Sf9-P122-1 |
| Negative control |
8.88% |
1.55% |
| 10-2 times dilution |
25.40% |
40.40% |
| 10-3 times dilution |
11.60% |
8.01% |
| 10-3 times dilution |
12.40% |
8.77% |
| 10-4 times dilution |
9.92% |
2.23% |
| 10-4 times dilution |
9.15% |
2.85% |
Table 2: Comparison of titer detection values of high passage cells (199 passages) and the cells which after three passage (122 passages).
Method verification
Versatility: The Futures Commission Merchant (FCM) method
can detect baculoviruses carrying different foreign genes, and the
method has a good versatility.
Repeatability: The same virus sample was tested repeatedly for
6 times, and the calculated RSD=1.58%, with good repeatability.
Intermediate precision: Three testers repeated the test for the
same virus sample three times, with RSD-average=1.41%, with
good intermediate precision.
Method application: Flow cytometry was used to detect the
titers of the seed bank, RBD and RBD dimer of the 2019-NCoV-S1
virus. There was no significant difference between the results of
flow cytometry and immune-fluorescence.
Discussion
The commercial kit (Expression Systems) of Futures Commission
Merchant (FCM) method uses Gp64-PE antibody for cell staining,
and the kit is restricted sales in some areas. In this article, Gp64-
APC antibody is used for detection. The concentration of this
antibody was 0.015 μg/test, which was lower than that of GP64-
PE antibody.
The titer value is calculated according to the virus dilution well,
which the positive cell proportion is about 10%, so there is no
need to draw a standard curve, so it is more efficient.
Unlike the plaque method, the Futures Commission Merchant
(FCM) method uses suspension culture, closely reflect actual
culturing conditions utilized for expression cultures, and can
obtain titer results within 24 hours. By using a centrifuge and a
flow cytometer equipped to handle 96-well plates, the Futures
Commission Merchant (FCM) method can easily achieve highthroughput
detection. Since the Futures Commission Merchant
(FCM) method detects the percentage of positive cells, it is not
necessary to analyze all cells to get the correct detection value,
during cell culture, centrifugation, cell staining, washing, etc.
even if the cell count is inaccurate, or the total cell volume is lost
due to cell shedding or other reasons, it will not affect the test
results.
Using vortex shaking can better resuspend the cells. Vigorous
vortex shaking will affect the cells that have bound antibodies
and reduce the detection value. But in the actual test, it was
found that the short-term vortex oscillation would not have a
significant impact on the test value. The reason may be that the
Gp64 antibody has a higher affinity for the antigen. The Futures
Commission Merchant (FCM) method has good reproducibility
for detecting virus titer. In addition to Futures Commission
Merchant (FCM) ability to accurately recognize the signal of a
single cell, the specific binding of Gp64 antibody to Gp64 also
plays a decisive role. Inactivation of Fasting blood sugar (FBS)
in diluent can reduce non-specific adsorption. Due to the good
specificity of binding of GP64 antibody to antigen, the content of
inactivated Fasting blood sugar (FBS) had no significant effect on
the detected value in the experiment.
The cell growth status had a significant effect on the titer value.
The operational process of cell passage and the cell cycle have
great influence on the cell growth state, which obviously affects
the titer detection value. In the operational process of multiple
successive passages, the cell state is prone to decline, which is
not suitable for titer detection. The poor cell growth status can
be adjusted by more rigorous cell passage operation, which is
suitable for titer detection again.
The grow cycle of Sf9 cell have four stages: adaptive phase,
logarithmic growth phase (early, middle, and late), plateau phase,
and recession phase. The density after passage has an effect on
the time of the cells to enter the logarithm phase. Generally
speaking, when the density is 1.0 ~1.2 × 106 cells/ml after
passage, the cells can enter the logarithmic growth phase within
24 hours. When the passage density is 0.8 ~ 1.0 × 106 cells/ml, the
adaptive phase is longer, and it will enter the logarithmic growth
phase after more than 24 hours. The cell state in the early phase
of the logarithm is the best, which is suitable for titer detection.
The detection values were lower when using mid logarithmic and
late logarithmic cells (Table 3). The cells of recession phase are
not suitable for titer detection (Table 4).
| Sample |
SF9/APC-A+ | Freq. of Parent |
| Sf9-P41-2 days |
Sf9-P54-2 days |
| Viable cell density:3.08 × 106 cells/ml |
Viable cell density:5.71 × 106 cells/ml |
| Negative control |
0.85% |
0.80% |
| 10-2 times dilution |
71.40% |
69.50% |
| 10-3 times dilution |
34.20% |
21.20% |
| 10-3 times dilution |
34.40% |
18.40% |
| 10-4 times dilution |
5.49% |
2.76% |
| 10-4 times dilution |
5.91% |
2.80% |
Table 3: Another virus was used to verify the influence of cell status on titer detection values.
| Baculovirus |
Titer-1 (107 ivp/ml) |
Titer-2 (107 ivp/ml) |
Titer-average (107 ivp/ml) |
| H5N1-HA |
49.4 |
53.6 |
51.5 |
| H5N1-NA |
4.5 |
4.38 |
4.44 |
| H5N1-M1 |
5.55 |
5.07 |
5.31 |
| H5N1-M2 |
4.13 |
4.18 |
4.16 |
| H5N1-NP |
2.86 |
2.88 |
2.87 |
| H1N1-HA |
16.8 |
17.6 |
17.2 |
| 2019-nCOV-N |
96.4 |
108 |
102.2 |
| 2019-nCOV-S |
58.1 |
58.7 |
58.4 |
| Empty baculovirus |
193 |
198 |
195.5 |
Table 4: Universality verification.
The assay is based on detection of the baculovirus gp64 fusion
protein which is expressed on the surface of infected insect
cells after infection (Table 5). Ensure that the incubation time is
between 13-22 hours, which is very important to obtain accurate
titer detection values. If the incubation time is less than 13 hours,
the GP64 protein cannot be adequately expressed and distributed
on the surface of infected cells, resulting in low detection value
(Table 6). If the incubation time is more than 22 hours, secondary
infection will occur (the progeny virus produced first, which then
infects healthy cells) (Table 7).
| Times of repetition |
Titer-1 (107 ivp/ml) |
Titer-2 (107 ivp/ml) |
Titer-average(107 ivp/ml) |
| 1 |
8.29 |
8.54 |
8.42 |
| 2 |
8.48 |
8.13 |
8.31 |
| 3 |
8.49 |
8.16 |
8.33 |
| 4 |
8.26 |
8.52 |
8.39 |
| 5 |
8.67 |
8.52 |
8.6 |
| 6 |
8.63 |
8.59 |
8.61 |
| Average |
|
|
8.44 |
| RSD(%) |
|
|
1.58 |
Table 5: Repeatability verification.
| Times of repetition |
The operator A-titer (107 ivp/ml) |
The operator B-titer (107 ivp/ml) |
The operator C-titer (107 ivp/ml) |
| 1 |
8.29 |
8.22 |
8.32 |
| 2 |
8.16 |
8.37 |
8.35 |
| 3 |
8.4 |
8.08 |
8.12 |
| Average |
8.28 |
8.22 |
8.26 |
| RSD (%) |
1.45 |
1.76 |
1.51 |
| RSD-Average (%) |
1.41% |
|
|
Table 6: The intermediate precision.
| Virus bank |
Titer-1 (107 ivp/ml) |
Titer-2 (107 ivp/ml) |
Titer average (107 ivp/ml) |
fluorescence immunoassay (107 PFU/ml) |
| S1-primary |
6.99 |
7.18 |
7.09 |
7.51 |
| S1-secondary |
28.7 |
26.1 |
27.4 |
38.6 |
| S1-working |
45.4 |
46.5 |
45.95 |
32.85 |
| RBD-primary |
16.6 |
13.3 |
14.95 |
18.42 |
| RBD-secondary |
26.8 |
27.2 |
27 |
24.57 |
| RBD-working |
36.6 |
33.3 |
34.95 |
28.62 |
| RBD dimer-primary |
47.5 |
48.2 |
47.85 |
42.4 |
| RBD dimer-secondary |
35.1 |
35.6 |
35.35 |
38.93 |
| RBD dimer-working |
54.9 |
59.1 |
57 |
58.76 |
Table 7: FCM method and fluorescence immunoassay were used to detect the titer of 2019-nCOV virus.
Conclusion
In addition, the doubling time of insect cells, It is 16 to 24 hours.
If the culture time is much longer than the doubling time, the
uninfected cells will expand, resulting in a noticeable change in
the total number of living cells in the culture well, which will also
affect the detection value.
References
- Wu Qingsheng, Li Yuanyuan (2020) Research progress on key technologies and applications of insect baculovirus expression vector system. Inter J Virolo 27:348-352.
- Yang J, WangW, Chen Z, Wei X, etal. (2020) A vaccine targeting the RBD of the S protein of SARS-CoV-2 induces protectiveimmunity.Nature 586:572-577.
- Wang Yan, Tan Feifei, Li Xiangdong, Tian Kegong (2018) Research progress on the determination of baculovirus titer [A]. Chinese Society of Animal Husbandry and Veterinary Medicine Branch of Veterinary Public Health. 6th Academic Session of Chinese Society of Animal Husbandry and Veterinary Medicine Symposium Proceedings [C]. Chinese Society of Animal Husbandry and Veterinary Medicine Branch of Veterinary Public Health: Chinese Society of Animal Husbandry and Veterinary Medicine Branch of Veterinary Public Health.
- Zhang Yuexia, Zhang Liying, Zhao Quan (2019) Current status and prospects of medical applications of flow cytometry. J Chinese Contemporary Medi 26:28-30.
- Sivaraman Divya, Yeh Hsiao-Yun, Mulchandani Ashok, Marylynn V Yates, Wilfred Chen (2013) Use of flow cytometry for rapid, quantitative detection of poliovirus-infected cells via TAT peptide-delivered molecular beacons. Appli Enviro Microbiolo 79:696-700.
- Long Yuyan, Guo Wang, Lan Huangli, Qu Dacai, Liang Xiang, et al. (2019) The research progress of silkworm nuclear polyhedrosis virus envelope glycoprotein GP64. J Guangxi Sericulture 56:25-32.
- Lin, Chung, Hu (2014) Update on baculovirus as an expression and/or delivery vehicle for vaccine antigens. 13:1501-1521.
- Mulvania T, Hayes B, Hedin D (2004) A Flow Cytometric Assay for Rapid, Accurate Determination of Baculovirus Titers. BioProcess J 3:47-53.