Research Article - (2018) Volume 4, Issue 2
Kwang Su Seo1*, Susan A Lettieri2, Steve K Henning2, Weng-Liang Hsu2, Adel F Halasa1*
1Institute of Polymer Science, The University of Akron, Akron, OH, USA
2The Goodyear Tire and Rubber Co., 142 Goodyear Blvd., Akron, OH, USA
*Corresponding Author:
Kwang Su Seo
Institute of Polymer Science
The University of Akron, Akron, OH, USA
Tel: +82-10-2378-0381
E-mail: mura1977@hotmail.com
Adel F. Halasa
Institute of Polymer Science
The University of Akron, Akron, OH, USA
Tel: +82-10-2378-0381
E-mail: halasa@uakron.edu
Received Date: July 09, 2018; Accepted Date: July 31, 2018; Published Date: August 06, 2018
Citation: Seo KS, Lettieri SA, Henning SK, Hsu WL, Halasa AF (2018) Determination of Reactivity Ratios for α-Methylstyrene- Butadiene Copolymerization via Cesiumbased Catalyst System. Polym Sci Vol.4 No.2:12
DOI: 10.4172/2471-9935.100040
The objective of this work is to determine reactivity ratios of the poly(α-methylstyrene-co-butadiene) polymerization in cesium-based catalyst system. These values can be used to describe the characteristic incorporation order of the monomers into the polymer chain and model the chain microstructure. The anionic solution polymerization of α-methylstyrene and 1,3-butadiene was polymerized at 10°C with different feed ratios in the presence of Cesium alkoxide, dialkyl magnesium, and chelating diamine. The reactivity ratio for butadiene at 50/50 wt% (35/65 molar ratio) α-methylstyrene/butadiene calculated as approximately 1.50, while that for α-methylstyrene calculated by both the integrated and differential methods were the values of 0.40 and -0.1. This would indicate that copolymer composition equation need to be modified to include propagation-depropagation equilibrium phenomenon.
Keywords
Copolymerization; Coplolymer; Polymerization; α-methylstyrene; Catalyst system
Introduction
The use of α-methylstyrene as a monomer has been limitedly applied to low temperature polymerization system due to the ceiling temperature of α-methylstyrene [1-4]. The restriction to low temperatures results from the thermodynamic equilibrium that favors a dimer over higher oligomers as the ceiling temperature of a-methylstyrene is approached [6-11]. The copolymerization of α-methylstyrene and conjugated dienes in hydrocarbon solvent at 65-75°C was studied using a novel new catalyst system based on organometallic compound of group I (n-butyl lithium) or group II (dibutyl magnesium) in combination with the alkoxides potassium or cesium of 2-etylhexyl alcohol in the presence of chelating diamine such as, N,N,N’,N’-tetramethylethylene diamine [TMEDA, 9]. The catalyst system based on the cesium alkoxide/TMEDA/ dibutyl magnesium in molar ratio 2:2:1 has previously proven that a comonomer mixture of α-methylstyrene and butadiene can be copolymerized at 65°C without the homopolymerization of α-methylstyrene [9]. The monomer of α-methylstyrene should be incorporated into the polymer chain as singlet units with the possibility of a small fraction of diads, since the rate of selfpropagation for α-methylstyrene is near zero in high temperature [10,11], the rate of cross-propagation must be higher. Therefore, α-methylstyrene can be fully converted into a polymer chain, restricting the negative rate effect of depropagation.
Determining the reactivity ratios for the copolymerization [12] of a-methylstyrene and 1,3-butadiene should provide further insight to the incorporation order outlined above. Differential methods and the integrated method of determining r1 and r2 based on the copolymer composition equation will be used to model the poly (α-methylstyrene-co-butadiene) rubber (α-MeSBR) copolymerization system.
Experimental
The anionic solution polymerization of α-methylstyrene (α-MeS) and 1,3-butadiene (Bd) was polymerized from 15wt% monomer concentration (315g monomer in 2100 total g hexane solution) in one-gallon reactor equipped with air driven motor stirrer nitrogen inlet and a cooling coil. Feed ratios of 10, 20, 30, 40, and 50 wt% α-MeS were used to polymerize the five α-MeSBRs used in the study. The reactions were run at 10°C to slow the kinetics enough to provide accurate conversion data. The catalyst consists of Cesium salt of 2-ethyl hexyl alcohol (6.3 mmol) reduced with n-butyllithium or dialkyl magnesium (3.15 mmol) in the presence of a chelating diamine (N, N, N’, N’ tetramethylethylene diamine, 6.3 mmol) to give final target molecular weight near 100,000 g/ mol. The reaction kinetics was followed by taken samples during the polymerization into a sealed glass vial containing undecane/ ethanol mixture using the undecane as an internal standard and analyzed for monomer consumption by a gas chromatography. At full conversion, the copolymer was terminated with ethanol and transferred to a vessel from the reactor to be stabilized with an antioxidant. The polymer was then dried in a vacuum oven. Reactivity ratios were calculated from experimental data. The monomer composition in copolymers was determined by 13C and 1H NMR.
Results and Discussion
The Kinetics of α-methylstyrene (α-MeS) copolymerizing with 1,3-butadiene (Bd) is unknown in the anionic polymerization system above the ceiling temperature of α-MeS. However, we have followed the assumption (r1=k11/k12) and (r2=k22/k21) whereas M1+ M2 form a chain end terminated with M2. Results of the calculations for determining the reactivity ratios r1 (Bd) and r2 (α-MeS) using both the integrated method and differential methods are shown in Table 1.
Table 1 Reactivity ratios of butadiene an α-methylstyrene.
| α-MeSBR composition | r1 (Bd) | r2 (α-MeS) | diff2 |
|---|---|---|---|
| Integrated Method | |||
| 10/90 | 1.81 | 2.64 | 0.0390 |
| 20/80 | 1.80 | 1.49 | 0.0103 |
| 30/70 | 1.70 | 0.73 | 0.0045 |
| 40/60 | 1.75 | 0.57 | 0.0072 |
| 50/50 | 1.47 | 0.39 | 0.0032 |
| Differential Methods R2 | |||
| Fineman/Ross | 1.60 | -0.09 | 0.996 |
| Kelen/Tudos | 1.55 | -0.09 | 0.997 |
There are considerable differences within each set of r1 and r2 values as calculated by the integrated method. As the weight % of α-MeS in the feed is increased, r1 and r2 appears to decrease with a value of 0.4 and 1.5, respectively. We believe that this phenomenon is a direct result of the original monomer feed ratio. At 10wt% α-MeS in feed, this value only corresponds to 6% molar quantity α-MeS, as seen in Table 2. It has been found that only a certain range of monomer feed ratios (molar) leads to the most reliable data suitable for calculating reactivity ratios; the range is 30-70% M2 [12]. Only the 50/50 wt% feed ratio falls in this range, equal to 35/65 molar ratio α-MeS/Bd. Therefore, only the r1, r2 values for this feed ratio can be considered valid. It is interesting to note that as the wt% of α-MeS in feed increases from 10% to 50%, both reactivity ratio values decrease. At lower molar amounts of α-MeS in system, the value of r1(Bd) may inflated due to a lower value for k12, the cross-propagation term, since there are low amounts of M2 (α-MeS) in system. The value of r2 (α-MeS) may be higher since lower concentrations of the monomer capable of depropagation may in turn artificially inflate its rate constant for forward propagation.
Table 2 Incorporated mol% of α-MeS and Bd in α-MeSBR analyzed by GC.
| Monomer feeding ratio (α-MeS/Bd) (wt% ratio) | α-MeS (mol%) | Bd (mol%) |
|---|---|---|
| 10/90 | 6 | 94 |
| 20/80 | 12 | 88 |
| 30/70 | 19 | 81 |
| 40/60 | 26 | 74 |
| 50/50 | 35 | 65 |
Using the differential methods developed to calculate r1, r2 values, different results were seen. The r1 values agree with those calculated from the integrated form of the copolymer composition equation for 50/50 wt%, but the values for r2 were not in agreement. The value of -0.10 can be considered as zero when considering errors inherent to sampling and calculations. The value of 1.5-1.6 for r1 (Bd) can be considered valid with respect to the mechanism of incorporation outlined in the background section of this publication. This value indicates that the butadienyl-Cs live end will show a slight preference to selfpropagation over cross-propagation to α-MeS monomer. The r2 value of 0.0-0.4 indicates either a very small tendency to selfpropagate or, in the case of r2=0.0, no self-propagation.
The resultant polymer would display a highly random incorporation tendency with small runs of Bd units dispersed by single α-MeS units. The polymer composition vs. total conversion data in Figure 1 for 50/50 w% α-MeSBR supports this conclusion, showing almost constant composition of α-MeS in the polymer chain above roughly 10% conversion. In addition, following the above proposed mechanism of comonomer incorporation, feed ratios of 50 wt% α-MeS and above do not incorporate higher than 45-48 w% α-MeS into chain at total conversion. This follows the assumption that the incorporation of the monomer capable of depropagation is dependent on the cross-propagation addition of the diene monomer. For example, in the extreme case of assuming a perfectly alternating copolymerization of higher than 50 mol% feed ratio α-MeS, the total amount of α-MeS polymerized will only equal that of the diene comonomer.

Figure 1: Time (minutes) vs. conversion (%) of α-MeSBR (30/70) via 2/2/1 of CsOR/TMEDA/MgBu2.
As the % of α-MeS in the initial feed ratio increases from 10% to 40% by weight (Table 3), the Tg of the resultant polymer increases due the proposed relatively random incorporation of α-MeS units into the chain. As shown in Figure 2 the polymers show relatively constant composition with conversion after approximately 10% conversion. The 50/50 α-MeSBR displays a Tg lower than what would be expected, following the above trend. In addition, NMR results show no peaks in the spectra that can be associated with hydrogen absorbance due to block α-MeS in the polymer [data not shown]. Preliminary results from ozonolysis and reduction analysis of the 40/60 α-MeSBR sample polymer indicate no large α-MeS blocks [9,10].
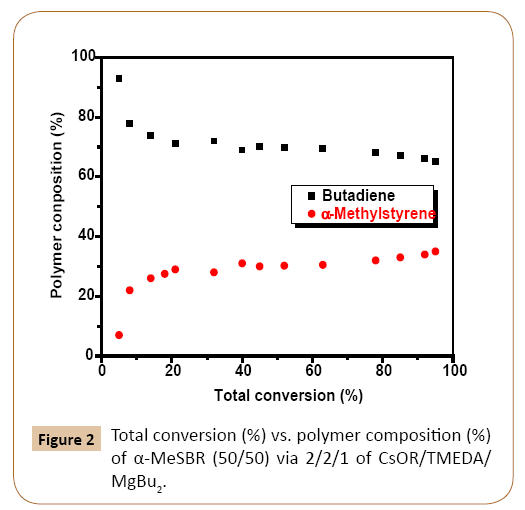
Figure 2: Total conversion (%) vs. polymer composition (%) of α-MeSBR (50/50) via 2/2/1 of CsOR/TMEDA/MgBu2.
Table 3 Monomer composition at 90% conversion characterized by NMR and its glass transition temperature.
| α-MeSBR Feed ratio | Tg (°C) | α-MeS (wt%) | Bd (wt%) |
|---|---|---|---|
| 10/90 | -49 | 11 | 89 |
| 20/80 | -40 | 21 | 79 |
| 30/70 | -30 | 29 | 71 |
| 40/60 | -22 | 37 | 63 |
| 50/50 | -33 | 45 | 55 |
The reactivity ratio values reported above are those calculated from equations derived under the assumption that all propagation steps are irreversible and that no depropagation at the live chain end exists. In this study, even though the reaction temperature is below the ceiling temperature for the neat α-MeS monomer, depopulation still exists because our amps monomer concentration is not high enough to eliminate depolymerization at the actual reaction temperature of 10°C [8]. In order to accurately calculate reactivity ratios from copolymerization’s involving one or more monomers that are capable of depolymerization at the chain end, the copolymer composition equation must be modified to include terms that account for propagation-depropagation equilibrium and contributions of the depropagation step to the forward rate constant [13]. The current methods utilized for calculating reactivity ratios in this analysis do not account for this phenomenon. It is possible for a depropagation step if an α-methylstyrenyl-Cs chain end is present and α-MeS monomer adds to this active center before a cross-propagation step can take place, creating a butadienyl-Cs center, then the reactivity ratios presented above may not accurately describe the incorporation of co-monomers into polymer.
Conclusion
Reactivity ratios of the poly(α-methylstyrene-co-butadiene) polymerization was studied in cesium-based catalyst system. At 50/50 wt% (35/65 molar ratio) α-MeS/Bd, the reactivity ratio for butadiene calculated by both the integrated method and differential methods agree at approximately 1.50. At the same feed ratio, the reactivity ratios for α-MeS calculated by both the integrated and differential methods are not in agreement, being 0.40 and -0.1, expecting to be zero for r2 (α-MeS). The differences in the above r2 values may stem from the complications involved with modeling a system exhibiting reversible propagation steps with equations based on solely irreversible propagation at the chain end.