Abdurrahman Sönmezler1* and Sakir Özgür Keskek2
1 Department of Neurology, Adana City Research and Training Hospital, Adana, Turkey
2 Department of Internal Medicine, Adana City Research and Training Hospital, Adana, Turkey
*Corresponding Author:
Abdurrahman Sönmezler
Assistant Professor
Department of Neurology
Adana City Research and Training Hospital, Adana, Turkey
Tel: +905323951977
Email: sonmezlera@gmail.com
Received Date: January 24, 2019; Accepted Date: February 26, 2019; Published Date: March 05 2019
Citation: Sönmezler A, Keşkek ÃƒÆ’Ã¢â‚¬Â¦Ãƒâ€šÃ…Â¾Ö (2019) Associated Microorganisms in clinical specimens from neurology intensive care unit of a tertiary care hospital in Adana, Turkey. J Intensive & Crit Care Vol.5 No.1:7
Objective: The aim of this study was to determine our microorganisms breeding in culture cause of nosocomial infections in neurology intensive care unit (NICU) of a tertiary care hospital.
Methods: This study was performed at the neurology intensive care unit, Adana City Research and Training Hospital, Turkey. A retrospective review of medical records was conducted for all patients admitted to the NICU, and clinical specimens of patients suspected of having infection were received, from September 2017 through July 2018. Data was collected using a neurology intensive care unit electronic database. MedCalc 15.8 software program (MedCalc Belgium) was used for statistical evaluation.
Results: Out of the 84 patients evaluated, there was no microbial growth in the clinical specimens of 24 (28.2%) patients. Microbial growths were observed in blood, urine or tracheal aspirate cultures of 60 (71.8%) of the patients, while there were microbial growths detected in blood cultures of 46 (54.1%) patients; urine cultures of 47 (55.27%) patients, and tracheal aspirate cultures of 15 (17.6%) patients. The highest percentage of bacterial recovery was Escherichia coli (23.6%), which were followed by Staphylococcus epidermidis (10.7%), Acinetobacter baumannii (9.5%), and Staphylococcus aureus (9.5%) respectively.
Conclusion: Clinical specimens from neurology intensive care unit of Adana City Research and Training Hospital, Turkey, yielded Gram-negative (Acinetobacter baumannii and Escherichia coli), as well as Gram-positive (Staphylococcus aureus and Staphylococcus epidermidis) bacterial species.
Keywords
Microorganisms; Microbial culture; Neurology intensive care unit
Introduction
Health care-related infection is an important factor in determining the clinical outcome among patients presenting to critical care areas. Surveillance of device-related infections has become an integral feature of infection control in all hospitals. Catheterrelated urinary tract infections, central line-related bloodstream infections, and ventilator-associated pneumonia are among the nosocomial infections commonly reported [1-6]. The Center for Disease Control and Prevention has provided simple definitions for the diagnosis of these infections [7], while any hospital's Infection Control Committee, serves as an important tool for monitoring of these infections.
In developed countries' hospitals produce infection control surveillance data, especially in the intensive care unit (ICU), as well as the epidemiology of antimicrobial resistance to such infection etiologic microorganisms, model, type, nature and risk factors, in cases where comprehensive information is needed [8]. Infections that are not present at the time of patient's admission to the hospital, but acquired while admitted in hospital, during hospitalization or after discharge are known as nosocomial infections, though in few cases, described as, Health Care Associated Infections (HCAI). These infections are important problem for health professionals, due to the increase in disability and mortality rates, economic losses, and the emergence of antimicrobial resistant microorganisms. It is seen in 5-10% of hospitalized patients [9].
Critically ill patients admitted in intensive care units (ICU) are always at a higher risk of developing nosocomial infections with resistant strains [10] because such patients have an increased susceptibility to infections of decreased mobility and increased use of invasive devices [11]. Infection with resistant strains in the ICUs leads to increased mortality and cost [12]. Nosocomial infections are particularly reported to be more pronounced in ICUs [13]. Thus, the aim of this study was to determine the microorganisms associated with clinical specimens of patients in neurology intensive care units (NICU), especially for reflective status in the Adana City Research and Training Hospital, Turkey, for the development of new decision-making and preventive infection control methods.
Materials and Methods
This retrospective study was performed in Adana City Hospital in NICU between September 2017 and July 2018. Patients who developed ICU infections after 48 hours from admission to ICU were included. The institutional review board of the hospital approved this experiment. All procedures were followed in accordance with the Good Clinical Practice standards and ethical standards of the Responsible Committee on Human Experimentation, and with the Helsinki Declaration of 1975, as revised in 2008. Patients who had signs of infection before being admitted to the ICU were excluded from the study. Blood, urine, tracheal aspirate, wound swab culture samples were collected from patients suspected of infection. Samples collected from the ICU unit were inoculated into 5% sheep blood agar and MacConkey agar plates and incubated overnight at 37°C aerobically. Bacterial pathogens were identified by conventional biochemical methods according to standard microbial techniques.
The MedCalc v18.5 software proGram (MedCalc Belgium) was used for statistical analysis. Data were reported as the mean ± standard deviation. Kolmogorov-Smirnov test was used to show the normal distribution of quantitative measurements. Chisquare is used to test the statistical significance of differences in sex distribution and smoking status. T test or Mann Whitney U tests were used for comparison of quantitative measurements. The probability of making a Type I error (alpha, significance) is 0.05 in all tests.
Results
Eighty four patients were evaluated in this retrospective study. The mean age was 68.1 ± 14.3. Female/Male ratio was 38/47. The mean age of the female patients was 71.2 ± 11.6 while it was 65.4 ± 15.8 in male patients. There was no statistical significant difference between the ages of males and females (p=0.065). The number of survived and died patient were 53 (63.5%) and 31 (36.5%), respectively. There was no breeding in the cultures of 24 patients (28.2%), blood, urine or tracheal aspirate culture breeding was observed in 60 patients (71.8%). There was breeding detected 46 patients’ (54.1%) blood cultures, 47 (55.27%) patients’ urine cultures, 15 patients’ (17.6%) tracheal aspirate cultures. The highest percentage (23.6%) was recorded for Escherichia coli, which was followed by Staphylococcus epidermidis (10.7%), Acinetobacter baumannii (9.5%), and Staphylococcus aureus (9.5%). Enterococcus faecalis (7.1%), Klebsiella pneumoniae (7.1%), Pseudomonas aeruginosa (5.9%), Staphylococcus capitis (5.9%), Candida albicans (5.9%). There were a higher percentage of Gram-negative bacilli than Grampositive bacilli (Supplementary Table 1). 31 (36.9%) patients were died, 53 (63.1%) patients were alive. There was no statistically significant difference between CRP levels of died and survived patients (8.9 ± 12.1 vs. 7.1 ± 10.0, p=0.641). The mean Glasgow coma scale of the dead patients was 5.9 ± 0.8 while it was 9.6 ± 0.8 in survivors. The difference was statistically significant (p<0.001) (Figure 1).
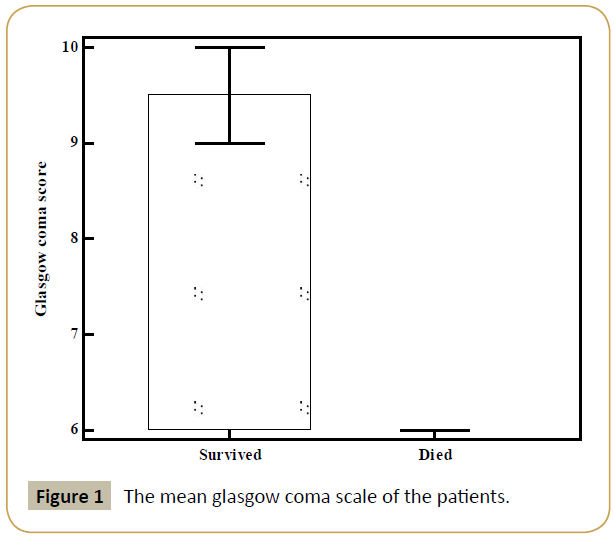
Figure 1: The mean glasgow coma scale of the patients.
Discussion
The aim of this study was to determine the microorganisms associated with clinical specimens of patients in neurology intensive care units (NICU), especially for reflective status in the our hospital. Hospital infections are one of the most important health problems in the world and in our country [14]. Patients with seventy years of age, shock, major injuries, acute renal failure, coma, and previous history of antibiotic use, drugs that suppress the patient's immune system, permanent catheters, and hospitalization for more than three days in ICU are important risk factors for HCAI [15]. The number of patients in the ICU, the low number of health personnel, the architectural structure of the intensive care unit, the lack of attention to hand washing, disinfection and sterilization, the failure to comply with the asepsis and isolation rules may also pave the way for the development of infection [16].
As the continent, country, city and hospital change, microorganisms that cause hospital infections change. In addition, the characteristics of patients, which are accepted to intensive care, are also important for infection. Studies report that the hospital infections in the ICU are predominantly caused by Gram negative bacteria [17,18]. In our study’ results were similar. The most commonly isolated organisms were gram-negative. In an international point prevalence study of 4947 infected ICU patients with microbiologic culture results from 75 countries, 62% of the positive isolates were Gram-negative organisms, 47% were Gram positive, and 19% were fungal. Staphylococcus aureus (20%) was the most common Gram-positive pathogen, whereas Pseudomonas spp (20%) and Escherichia coli (16%) were the most common Gram-negative pathogens reported in patients (18) the study in ICUs from both the United States and Europe found that Escherichia coli, Klebsiella spp., Pseudomonas aeruginosa, and Enterobacter spp. are the 4 most frequently isolated gramnegative organisms. Among ICU-associated Gram-negative bacterial pathogens in the United States, Klebsiella pneumoniae followed by E coli was the most prevalent [19]. Al Johani et al. [20] reported that of 4192 isolates taken from clinical specimens between 2004 and 2009, 2792 (66.6%) were gram-negative organisms and 1400 (33.4%) were gram-positive organisms.
Qadeer et al. [21] observed that the predominant gram-negative isolates in their study were Acinetobacter and Escherichia coli. Al-Jawady, et al. [22] and Morfin-Otero, et al. [23] concluded that Escherichia coli were the most common Gram-negative organism being isolated from the ICU. Qadeer et al. [21] observed that the most frequent isolate from urine was found to be Escherichia coli in their study. Urinary tract infection (UTI) is the most common infection in developing countries UTIs are caused by blood and pathogens that enter the urinary tract through the perineum, digestive tract or urinary catheter [24,25]. Doyle et al. [26] concluded that morbidity and mortality are significant causes of infections in ICU; multi resistant Gram-negative bacilli were reported to be most prevalent germs responsible for infections. Xie et al. [27] reported that Pseudomonas aeruginosa, Escherichia coli, and A. baumannii as the most prevalent germs consistent with nosocomial infection. Esfahani et al. [28] concluded that most prevalent bacteria causing infections are Pseudomonas aeruginosa, Klebsiella spp., and Escherichia coli. Most studies report that more than half of the hospital infections in the ICU are caused by Gram negative bacteria [29]. Deng et al. [30] concluded that the most common organisms causing blood stream infections were coagulase-negative Staphylococci (44.6% of isolates), Staphylococcus aureus (17.4%), Klebsiella spp. (11.5%), and Acinetobacter spp. (11.5%) but, the majority of the their patients had head trauma and surgical procedures. Thus, the study of only the bloodstream infection and predominance of surgical patients may explain Gram positive dominance. Lee et al. [31] concluded that hospital-acquired pneumonia due to Gram-negative pathogens has recently increased, especially in ICU settings. Our study found a high percentage of Gram-negative bacilli than gram-positive bacilli. The main cause of NICU infection was urinary tract infection (UTI) caused by Escherichia coli which was followed by Staphylococcus epidermidis, Acinetobacter baumannii, Staphylococcus aureus. These microorganisms are among the major causes of hospital infections worldwide. Despite all protective measures, nosocomial infections in our neurology intensive care unit, indicates that new and different decision-making and preventive methods should be taken to control infection.
References
- Jamoussi A, Ayed S, Ben Ismail K, Chtara K, Bouaziz M, et al. (2018) The prevalence of healthcare-associated infection in medical intensive care units in Tunisia. Results of the multi-centre nosorea1 study. Tunis Med 96: 731-736.
- Nazarchuk OA, Dmytriiev DV, Dmytriiev KD, Nazarchuk HH, Zaletskiy BV (2018) Characteristics of infectious complications in critically ill patients. Wiad Lek 71: 1784-1792.
- Ferreira RL, da Silva BCM, Rezende GS, Nakamura-Silva R, Pitondo-Silva A, et al. (2018) High Prevalence of Multidrug-Resistant Klebsiella pneumoniae Harboring Several Virulence and β-Lactamase Encoding Genes in a Brazilian Intensive Care Unit. Front Microbiol 9: 3198.
- Rai A, Khera A, Jain M, Krishnakumar M, Sreevastava DK (2019) Bacterial colonization of peripheral intravenous cannulas in a tertiary care hospital: A cross sectional observational study. Med J Armed Forces India 75: 65-69.
- Bonnet V, Dupont H, Glorion S, Aupée M, Kipnis E, et al. (2019) Influence of bacterial resistance on mortality in intensive care units: A registry study from 2000 to 2013 (IICU Study). J Hosp Infect pii: S0195-6701(19)30013-1.
- Su LX, Wang XT, Pan P, Chai WZ, Liu DW, et al. (2019) Infection management strategy based on prevention and control of nosocomial infections in intensive care units. Chin Med J (Engl) 132: 115-119.
- Horan TC, Andrus M, Dudeck MA (2008) CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 36: 309-332.
- Haley RW, Culver DH, White JW, Morgan WM, Emori TG, et al. (1985) The efficacy of infection surveillance and control programs in preventing nosocomial infections in US hospitals. Am J Epidemiol 121: 182-205.
- Khan HA, Baig FK, Mehboob R (2017) Nosocomial infections: Epidemiology, prevention, control and surveillance. Asian Pacific Journal of Tropical Biomedicine 7: 478-482.
- Singh AK, Sen MR, Anupurba S, Bhattacharya P (2002) Antibiotic sensitivity pattern of the bacteria isolated from nosocomial infections in ICU. J Commun Dis 34: 257-263.
- Rice LB (2003) Controlling antibiotic resistance in the ICU: Different bacteria, different strategies. Cleve Clin J Med 70: 793-800.
- Kaul S, Brahmadathan KN, Jagannati M, Sudarsanam TD, Pitchamuthu K, et al. (2007) One year trends in the gram-negative bacterial antibiotic susceptibility patterns in a medical intensive care unit in South India. Indian J Med Microbiol 25: 230-235.
- Cassini A, Plachouras D, Eckmanns T, Abu Sin M, Blank HP, et al (2016) Burden of six Healthcare-associated infections on European population health: estimating incidence-based disability-adjusted life years through a population prevalence-based modelling study. PLoS Med 13: e1002150.
- Brawley RL, Weber DJ, Samsa GP, Rutala WA (1989) Multiple nosocomial infections. An incidence study. Am J Epidemiol 130: 769-780.
- Mehta Y, Gupta A, Todi S, Myatra SN, Samaddar DP, et al. Guidelines for prevention of hospital acquired infections. Indian J Crit Care Med 18: 149-163.
- Spencer RC (1994) Epidemiology of infection in ICU’s. Intensive Care Med 20: 2-6.
- Richards MJ, Edwards JR, Culver DH, Gaynes RP (2000) Nosocomial infections in combined medical-surgical intensive care units in the United States. Infect Control Hosp Epidemiol 21: 510-515.
- Vincent JL, Rello J, Marshall J, Silva E, Anzueto A, et al. (2009) International study of the prevalence and outcomes of infection in intensive care units. JAMA 302: 2323-2329.
- Sader HS, Farrell DJ, Flamm RK, Jones RN (2014) Antimicrobial susceptibility of Gram-negative organisms isolated from patients hospitalized in intensive care units in United States and European hospitals (2009-2011). Diagn Microbiol Infect Dis 78: 443-448.
- Al Johani SM, Akhter J, Balkhy H, El-Saed A, Younan M, et al. (2010) Prevalence of antimicrobial resistance among gram-negative isolates in an adult intensive care unit at a tertiary care center in Saudi Arabia. Ann Saudi Med 30: 364-369.
- Qadeer A, Akhtar A, Ain QU, Saadat S, Mansoor S, et al. (2016) Antibiogram of Medical Intensive Care Unit at Tertiary Care Hospital Setting of Pakistan. Cureus 8: e809.
- Al-Jawady ZA, Al-Habib HM (2012) Antibiogram profiles of bacterial isolates from intensive care units in Mosul Teaching Hospitals. Raf J Sci 23: 52-59.
- Morfin-Otero R, Tinoco-Favila JC, Sader HS, Salcido-Gutierrez L, Perez-Gomez HR, et al. (2012) Resistance trends in gram-negative bacteria: surveillance results from two Mexican hospitals, 2005-2010. BMC Res Notes 5: 277.
- Tessema B, Kassu A, Mulu A, Yismaw G (2007) Predominant isolates of urinary tract pathogens and their antimicrobial susceptibility patterns in Gondar University Teaching Hospital, northwest Ethiopia. Ethiop Med J 45: 61-67.
- Grabe M, Bjerklund-Johansen T, Botto H, et al. Guidelines on urological infections. European Association of Urology. 2015: 7-8.
- Doyle JS, Buising KL, Thursky KA, Worth LJ, Richards MJ (2011) Epidemiology of infections acquired in intensive care units. Semin Respir Crit Care Med 32: 115-138.
- Xie DS, Xiong W, Xiang LL, Fu XY, Yu YH, et al. (2010) Point prevalence surveys of healthcare-associated infection in 13 hospitals in Hubei Province, China, 2007-2008. J Hosp Infect 76: 150-155.
- Esfahani BN, Basiri R, Mirhosseini SMM, Moghim S, Dolatkhah S (2017) Nosocomial Infections in Intensive Care Unit: Pattern of Antibiotic-resistance in Iranian Community. Adv Biomed Res 6: 54.
- Dasgupta S, Das S, Chawan NS, Hazra A (2015) Nosocomial infections in the intensive care unit: Incidence, risk factors, outcome and associated pathogens in a public tertiary teaching hospital of Eastern India. Indian J Crit Care Med 19: 14-20.
- Deng S, Feng S, Wang W, Zhu H, Gong Y (2019) Bacterial Distribution and Risk Factors of Nosocomial Blood Stream Infection in Neurologic Patients in the Intensive Care Unit. Surg Infect (Larchmt) 20: 25-30.
- Lee HS, Moon J, Shin HR, Ahn SJ, Kim TJ, et al. (2019) Pneumonia in hospitalized neurologic patients: trends in pathogen distribution and antibiotic susceptibility. Antimicrob Resist Infect Control 8: 25.