Michele Figliuzzi1, Amerigo Giudice1, Francesco Guido Mangano2,3* and Leonzio Fortunato1
1Department of Health Sciences, Dental School, Magna Grecia University, 88100 Catanzaro, Italy
2Department of Surgical and Morphological Science, Dental School, University of Insubria, 21100 Varese, Italy
3Academic Unit of Digital Dentistry, IRCSS San Raffaele Hospital, 20132 Milan, Italy
*Corresponding Author:
Francesco Mangano
Department of Surgical and Morphological Science, Dental School, University of Insubria, 21100 Varese, Italy
Tel: +390332393668
E-mail: francescoguidomangano@gmail.com
Received date: March 04, 2016; Accepted date: March 22, 2016; Published date: March 25, 2016
Citation: Figliuzzi M, Giudice A, Mangano FG, et al. A Direct Metal Laser Sintering (DMLS) Root Analogue Implant Placed in the Anterior Maxilla: Case Report. Periodon Prosthodon. 2016, 2:1. doi: 10.21767/2471-3082.100011
Keywords
Immediate implant; Root analogue implant; Titanium; Direct metal laser sintering
Introduction
Immediate implant placement is the insertion of an implant immediately after surgical extraction of the tooth to be replaced [1,2]. This procedure reduces treatment time and cost, with less surgical sessions and therefore a positive impact on the patient [1-3]; in addition, it may help to place the fixture in an ideal 3D position, and to enhance tissue maintenance, counteracting alveolar ridge contraction following tooth removal, at least to some extent [4,5]. However, immediate implant placement has disadvantages too: when an implant is placed in a fresh extraction socket, it can be difficult to obtain sufficient primary stability [6,7], because of the discrepancy between the fixture and the socket [2,3,6,7]. Primary stability is usually obtained by placing tapered implants, exceeding the apex of the socket by 2-4 mm, or using wide diameter implants [2,3,6,7]. One possible alternative can be the fabrication of a custom-made, rootanalogue implant (RAI) [8,9]. Several techniques of fabricating and placing custom-made RAI have been noted in the literature [8-14]; recently, however, modern additive manufacturing (AM) technologies, including direct metal laser sintering (DMLS), have opened new interesting perspectives for the fabrication of custom-made RAIs [8,9,15]. With DMLS, a powerful laser beam is directed on a bed of titanium micro-powders, and fuse them in accordance to a computer-assisted-design (CAD) file, in order to generate thin metal layers [16,17]. Layer-by-layer, the desired 3D object is finally fabricated, without post-processing [16,17]. Several human histologic/ histomorphometric studies have documented the bone response after the insertion of DMLS titanium implants [18,19]; the clinical performance of DMLS titanium dental implants has been investigated, and satisfactory outcomes have been reported [20-23]. Today, DLMS can be used for the fabrication of custom-made, RAIs. In fact, modern cone beam computed tomography (CBCT) acquisition and 3D image conversion, combined with DLMS, allows the fabrication of custom-made RAIs, perfect copies of the radicular units to be replaced [8,9,15]. Although DMLS has proven to be an effective technique for manufacturing of custom-made RAIs [15], there is still limited clinical data in dental literature [9,24,25]; to date, there are no studies reporting on these implants in the esthetic area of the anterior maxilla. This report demonstrates how DMLS technology permits to fabricate customized titanium RAI, which can be immediately inserted and restored in a fresh extraction socket of the anterior maxilla.
Materials and Methods
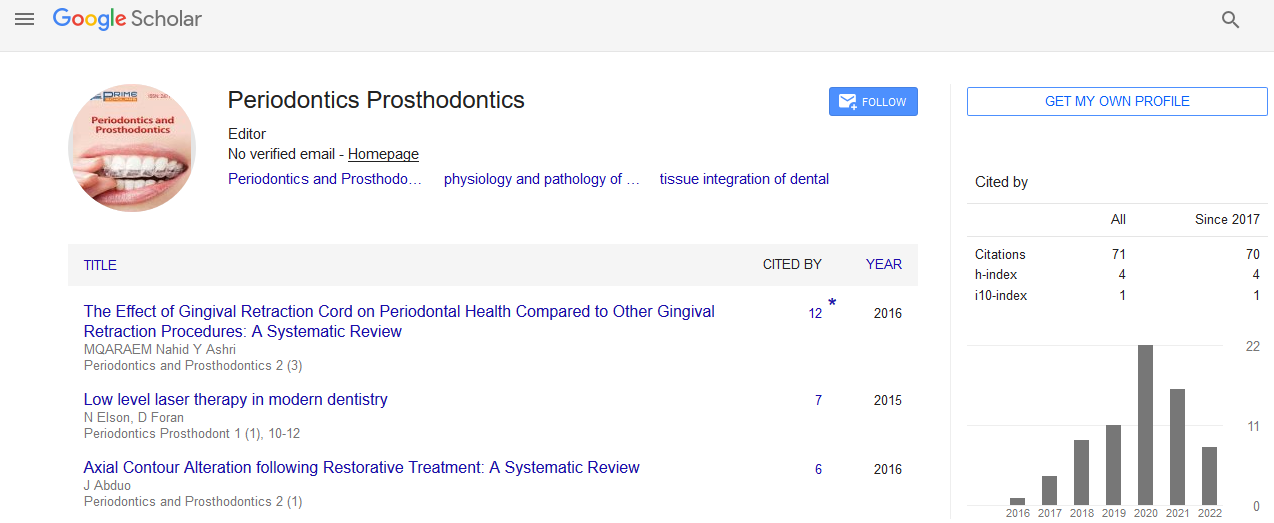
A 45-year-old male patient with a fractured non-restorable lateral right maxillary incisor was selected for the present study. The procedure for the fabrication of the RAI was as previously described [9,24,25]. The patient underwent a CBCT scan (CS9300®, Carestream Health, Rochester, NY, USA). DICOM datasets were then sent to a 3D reconstruction software (Mimics®, Materialise, Leuven, Belgium). With this software, segmentation was performed and a 3D recostruction of the maxilla and the residual, non-restorable root were obtained (Figure 1). A “virtual extraction” was performed, isolating the root as a stereolithographic (STL) file; this file was therefore sent to a reverse-engineering software (ProEngineering CAD 3D®, PTC Group, Needham, MA, USA), where the root was processed and the prosthetic abutment was added. The diameter of the implant neck was then reduced in the area in contact with the thin buccal bone, in order to protect if from pressure-induced resorption: this vestibular slice was obtained using a powerful software (Magics®, Materialise, Leuven, Belgium) (Figure 2). The RAI was fabricated via DMLS, using Ti–6Al–4V alloy powder, with a particle size of 25-45 micro-meters as the basic material, as previously described [9]. The patient was fully informed about the treatment modalities and signed an informed consent form. The study respected the principles outlined in the Declaration of Helsinki on experimentation involving human subjects (2008). Surgical and prosthetic procedure was as previously described [9]. After local anaesthesia with articaine 4% (containing 1:100,000 adrenaline) an intrasulcular incision was performed, and the non-restorable incisor was gently extracted, in order to avoid any damage to the socket. In fact, in the event of a loss of the buccal bone wall, primary stability of the RAI would be compromised, jeopardizing the treatment outcome. After tooth extraction, the socket was carefully debrided. Then, the RAI was gently inserted in the socket using a little percussion hammer. Primary stability was achieved, as a consequence of the congruence between the RAI and the socket, and carefully checked (Figure 3). Then, sutures were positioned. The RAI was immediately provisionalized with a cemented single crown, for esthetic reasons; care was taken, however, to avoid any functional contacts (Figure 4). Oral antibiotics were prescribed, amoxicillin+clavulanic acid, 2 g/d for 6 days (Augmentin®, GlaxoSmithKline Beecham, Brentford, UK) together with analgesics, 100 mg of nimesulide (Aulin®, Roche Pharmaceutical, Basel, Switzerland) every 12 hours for 2 days. Mouth rinses with 0.12% chlorexidine (Chlorhexidine®, Oral-B, Boston, MA, USA) were prescribed daily, for 7 days. The patient was seen on a weekly basis during the first month. At 1 week, no post-operative discomfort or edema was reported, and sutures were removed. At 2 weeks, the peri-implant tissues were stable, although not mature enough to avoid an esthetically unpleasing gray effect (Figure 5). The provisional restoration remained in situ for 3 months, after that the final metal-ceramic crown was delivered (Figure 6), and cemented with cemented with zinceugenol oxide cement (TempBond®, Kerr, Orange, CA, USA).
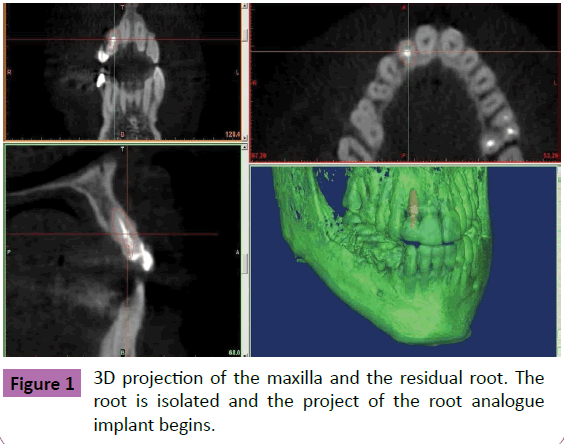
Figure 1 3D projection of the maxilla and the residual root. The root is isolated and the project of the root analogue implant begins.

Figure 2 The STL file of the custom-made RAI. An integral abutment has been added. The root analogue has been designed with a reduction of the diameter of the implant neck of 0.1-0.3 mm next to the buccal cortical bone.
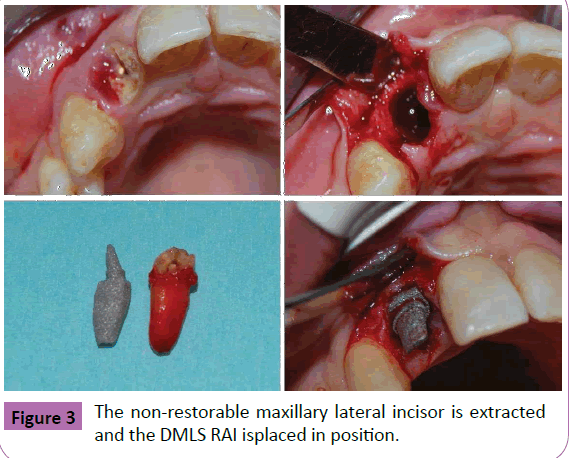
Figure 3 The non-restorable maxillary lateral incisor is extracted and the DMLS RAI isplaced in position.
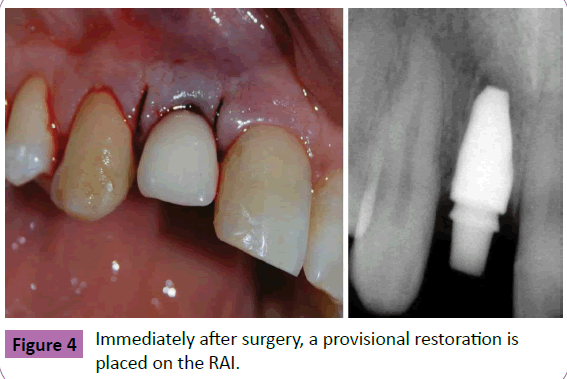
Figure 4 Immediately after surgery, a provisional restoration is placed on the RAI.
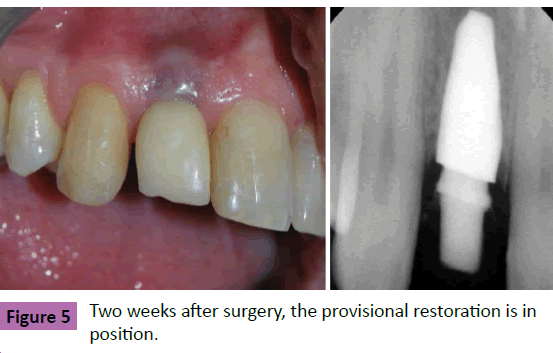
Figure 5 Two weeks after surgery, the provisional restoration is in position.
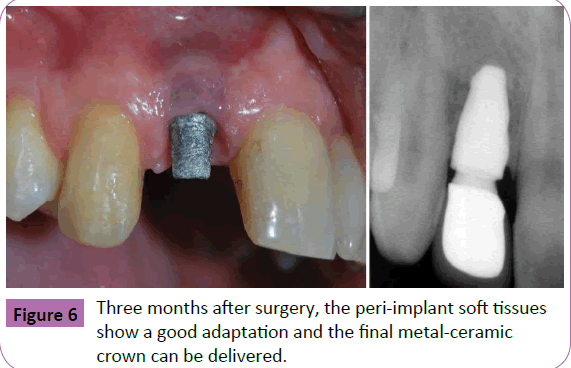
Figure 6 Three months after surgery, the peri-implant soft tissues show a good adaptation and the final metal-ceramic crown can be delivered.
Results
At the 1-year follow-up control, the RAI was successfully in function. No biologic complications were reported. The peri-implant tissues were mature and stable, and the esthetic integration of the crown was satisfactory. The radiographic control revealed little or no peri-implant bone loss, and no soft tissue recession was present. Radiographically, the implant-crown complex had a natural appearance, close to that of a natural tooth (Figure 7). No prosthetic complications were registered.
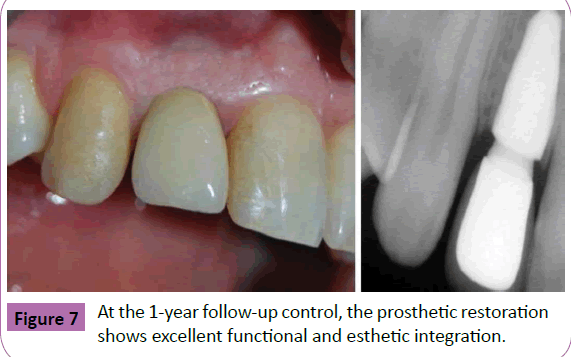
Figure 7 At the 1-year follow-up control, the prosthetic restoration shows excellent functional and esthetic integration.
Discussion
Primary stability is important, but it represents a challenge in fresh extraction sockets [2,3,6,7]. The fundamental pre-requisites for achieving primary implant stability include adequate bone quantity and quality, gentle extraction, and a careful preservation of the alveolar socket walls [2,3,6,7]. Primary stability is also related to the macroscopical features of the fixture (shape, length and diameter): in fact, it is usually achieved by inserting tapered implants, longer implants exceeding the alveolar apex, or wider implants [2,3,6,7]. As primary stability is a critical factor for osseointegration and long-term implant success, the use custommade, RAIs congruent to the individual extraction sockets may represent an interesting alternative treatment option [8,9]. The concept is not new, as the oldest evidence of RAI dates back to around 600 BC. While excavating Mayan burial sites in Honduras in 1931, the archaeologist Wilson Popenoe found a fragment of mandible of a young Mayan woman, with three tooth-shaped pieces of shell inserted to replace three missing lower incisors. The ancient Etruscans carved wood, metal, shell, and stones to form RAIs. In 1969, the polymethacrylate RAI used by Hodosh and coll did not osseointegrate [10]. In animal studies, titanium RAI showed signs of osseointegration [11,12]; however, the placement of these implants could fracture the thin buccal wall of the alveolar bone [11,12], as confirmed by a subsequent human study, where the failure rate was high [26]. The authors speculated that the perfect implant adaptation to the socket could be responsible for the intermediate-term failure, because of the subsequent uniform pressure-induced resorption concerning the entire alveolar surface [26]. Following single-tooth extraction, the alveolar bone generally exhibits remarkable modifications in its vertical and horizontal dimension: in particular, the bucco-lingual dimensions of the ridge can be severely reduced, with up to 50% reduction of the original ridge width [27-29]. A recent systematic review on clinical studies by Tan et al. has confirmed that after tooth extraction a pronounced horizontal dimensional reduction (3.79 ± 0.23 mm) combined with a vertical reduction (1.24 ± 0.11 mm on buccal, 0.84 ± 0.62 mm on mesial and 0.80 ± 0.71 mm on distal sites) occur at 6 months, and percentage horizontal and vertical dimensional changes are comprised between 29- 63% and 11-22% at 6 months, respectively [30]. The amount of bone resorption is usually greater at the buccal aspect than at its palatal/lingual counterpart [27-30], particularly in the anterior maxilla. In fact, most tooth sites in the anterior maxilla exhibit very thin buccal bone walls that are frequently made up of only bundle bone [31-33]. As in the anterior maxilla the bundle bone is a tooth-dependent structure with no or few blood vessels, such a thin wall usually undergoes marked resorption following tooth extraction [28,31,32], and it is also prone to pressure-induced resorption. In this context, a perfect fit of the implant in this area might be responsible for the esthetic failure because of pressure induced resorption [27]. In a series of studies, the immediate insertion of zirconia RAIs with a diameter reduction of 0.1-0.3 mm in the area in contact with the buccal bone and retentions in the interdental space yielded excellent functional and esthetic results [13,14,34]. No bone resorption or soft tissue recession was reported for these implants [13,14,34]. The reduction of the implant diameter with a slice in proximity of the buccal bone may avoid pressure-induce bone loss and related esthetic failure; the retentions in the interdental space may improve primary implant stability [13,14,34]. More recently, a novel approach to fabricate titanium custom-made RAIs using CBCT and DMLS has been proposed [9,15,24,25]. In our present study, a customized DMLS titanium RAI was fabricated and inserted into a fresh extraction socket of the anterior maxilla. The congruence between the RAI and the socket was excellent. The neck of the implant presented a diameter reduction of 0.1-0.3 mm, and a slice designed to prevent any pressure-induced bone loss in the critical buccal bone area, where aesthetics are the major concern [35]. At the 1-year followup control, the DMLS RAI showed a satisfactory integration, from the functional and esthetic point of view, with no bone resorption or soft tissue recession. The fabrication of custom-made RAI with the DLMS technique presents two possible advantages. On the one hand, the DLMS technique permis the fabrication of functionally graded titanium implants, with a highly porous surface and a dense core [16-23]. This may help to avoid any stress-shielding effect, further reducing pressure induced bone loss [16-23]. On the other hand, a porous surface is obtained through the DMLS process: this surface is capable to accelerate the healing processes and to finally promote Osseo integration [16-23].
Conclusions
In the last years, dentistry is evolving towards digitalization, in order to simplify clinical procedures and shorten treatment times. Nowadays, modern CBCT acquisition and 3D image processing, combined with the DMLS process, allows us to fabricate customized RAIs, In the present study, a custom-made DMLS titanium RAI was fabricated and inserted into a fresh extraction socket of the esthetic area of the anterior maxilla. After 1 year of follow-up, the integration of the DMLS implant was functionally and aesthetically satisfactory, with no bone resorption or soft tissue recessions. The fabrication and placement of customized titanium RAIs may represent a new fascinating option for immediate implantation.
Conflict of Interest
We declare no conflict of interest for this work.
References
- Chrcanovic BR, Albrektsson T, Wennerberg A (2015) Dental implants inserted in fresh extraction sockets versus healed sites: a systematic review and meta-analysis. J Dent 43: 16-41.
- Koh RU, Rudek I, Wang HL (2010) Immediate implant placement: positives and negatives. Implant Dent 19: 98-108.
- Ritter L, Palmer J, Bindl A, Irsen S, Cizek J, et al. (2014) Accuracy of chairside-milled CAD/CAM drill guides for dental implants. Int J Comput Dent 17: 115-124.
- Evans CD, Chen ST (2008) Esthetic outcomes of immediate implant placements. Clin Oral Implants Res 19: 73-80.
- Mangano FG, Mangano C, Ricci M, Sammons RL, Shibli JA, et al. (2013) Esthetic evaluation of single-tooth morse taper connection implants placed in fresh extraction sockets or healed sites. J Oral Implantol 39: 172-181.
- Bindl A, Ritter L, Mehl A (2010) Digital 3D implant planning: Cerec meets Galileos. Int J Comput Dent 13: 221-231.
- Javed F, Ahmed HB, Crespi R, Romanos GE (2013) Role of primary stability for successful osseointegration of dental implants: Factors of influence and evaluation. Interv Med Appl Sci 5: 162-167.
- Moin DA, Hassan B, Mercelis P, Wismeijer D (2013) Designing a novel dental root analogue implant using cone beam computed tomography and CAD/CAM technology. Clin Oral Implants Res 24: 25-27.
- Figliuzzi M, Mangano F, Mangano C (2012) A novel root analogue dental implant using CT scan and CAD/CAM: selective laser melting technology. Int J Oral MaxillofacSurg 41: 858-862.
- Hodosh M, Povar M, Shklar G (1969) The dental polymer implant concept. J Prosthet Dent 22: 371-380.
- Lundgren D, Rylander H, Andersson M, Johansson C, Albrektsson T (1992) Healing-in of rootanalogue titanium implants placed in extraction sockets. An experimental study in the beagle dog. Clin Oral Implants Res 3: 136-144.
- Kohal RJ, Hurzeler MB, Mota LF, Klaus G, Cafessee RG, et al. (1997) Custom-made rootanalogue implants placed into extraction sockets. An experimental study in monkeys. Clin Oral Implants Res 8: 386-392.
- Pirker W, Kocher A (2008) Immediate, non-submerged, root analogue zirconia implant insingle tooth replacement. Int J Oral MaxillofacSurg 37: 293-295.
- Pirker W, Wiedemann D, Lidauer A, Kocher A (2011) Immediate, single stage, truly anatomiczirconia implant in lower molar replacement: a case report with 2.5 years follow-up. Int J Oral Maxillofac Surg 40: 212-216.
- Moin DA, Hassan B, Parsa A, Mercelis P, Wismeijer D (2014) Accuracy of preemptively constructed, cone beam CT, and CAD/CAM technology-based, individual Root Analogue Implant technique: an in vitro pilot investigation. Clin Oral Implants Res 25: 598-602.
- Traini T, Mangano C, Sammons RL, Mangano F, Macchi A, et al. (2008) Direct laser metal sintering as a new approach to fabrication of an isoelastic functionally graded material for manufacture of porous titanium dental implants. Dent Mater 24: 1525-1533.
- Mangano F, Chambrone L, Noort VR, Miller C, Hatton P, et al. (2014) Direct metal laser sintering titanium dental implants: a review of the current literature. Int J Biomaterpp: 1-11.
- Mangano C, Piattelli A, Avila S, Iezzi G, Mangano F, et al. (2010) Early human bone response to laser metal sintering surface topography: a histologic report. J Oral Implantol 36: 91-96.
- Mangano C, Piattelli A, Raspanti M, Mangano F, Cassoni A, et al. (2011) Scanning electron microscopy (SEM) and X-ray dispersive spectrometry evaluation of direct laser metal sintering surface and human bone interface: a case series. Lasers Med Sci 26: 133-138.
- Mangano C, Mangano F, Shibli JA, Luongo G, Franco M, et al. (2012) Prospective clinical evaluation of 201 direct laser metal forming implants: results from a 1-year multicenter study. Lasers Med Sci 27: 181-189.
- Mangano C, Mangano FG, Shibli JA, Ricci M, Perrotti V, et al. (2012) Immediate loading of mandibular overdentures supported by unsplinted direct laser metal-forming implants: results from a 1-year prospective study. J Periodontol 83: 70-78.
- Mangano F, TaubertPS, Zecca PA, Luongo G, Sammons RL, et al. (2013) Immediate restoration of fixed partial prostheses supported by one-piece narrow-diameterselective laser sintering implants: a 2-year prospective study in the posterior jaws of 16 patients. Implant Dent 22: 388-393.
- Mangano FG, Caprioglio A, Levrini L, Farronato D, Zecca PA, et al. (2015) Immediate loading of mandibular overdentures supported by one-piece, direct metal laser sintering mini-implants: a short-term prospective clinical study. J Periodontol 86: 192-200.
- Mangano FG, Cirotti B, Sammons RL, Mangano C (2012) Custom-made, root-analogue direct laser metal forming implant: a case report. Lasers Med Sci 27: 1241-1245.
- Mangano FG, Franco M, Caprioglio A, Macchi A, Piattelli A, et al. (2014) Immediate, non-submerged, rootanalogue direct laser metal sintering (DLMS) implants: a 1-year prospective study on 15 patients. Lasers Med Sci 29: 1321-1328.
- Kohal RJ, Klaus G, Strub JR (2002) Clinical investigation of a new dental immediate implant system. The reimplant-system. DtschZahnarztl Z 57: 495-497.
- Botticelli D, Berglundh T, Lindhe J (2004) Hard-tissue alterations following immediate implant placement in extraction sites. J Clin Periodontol; 31: 820-828.
- Araujo MG, Silva CO, Misawa M, Sukekava F (2015) Alveolar socket healing: what can we learn? Periodontol 2000 68: 122-134.
- Farmer M, Darby I (2014) Ridge dimensional changes following single-tooth extraction in the aesthetic zone. Clin Oral Implants Res 25: 272-277.
- Tan WL, Wong TL, Wong MC, Lang NP(2012) A systematic review of post-extractional alveolar hard and soft tissue dimensional changes in humans. Clin Oral Implants Res 23: 1-21.
- Huynh-Ba G, Pjetursson BE, Sanz M, Cecchinato D, Ferrus J, et al. (2010) Analysis of the socket bone wall dimensions in the upper maxilla in relation to immediate implant placement. Clin Oral Implants Res 21: 37-42.
- Januario AL, Duarte WR, Barriviera M, Mesti JC, Araujo MG, et al. (2011) Dimension ofthe facial bone wall in the anterior maxilla: a cone-beam computed tomography study. ClinOral Implants Res 22: 1168-1171.
- Chappuis V, Engel O, Reyes M, Shalim K, Nolte LP, et al. (2013) Ridge alterations post-extraction in the esthetic zone: a 3D analysis with CBCT. J Dent Res 92: 195-201.
- Pirker W, Kocher A(2009) Immediate, non-submerged, root-analogue zirconia implantsplaced into single-rooted extraction sockets: 2-year follow-up of a clinical study. Int J Oral Maxillofac Surg 38: 1127-1132.
- Zimmermann M, Mehl A (2015) Virtual smile design systems: a current review. Int J Comput Dent 18: 303-317.